Continuing Part 1 with three DIM studies, the first of which was a 2020 chemical analysis investigating:
“Anti-estrogenic, anti-androgenic, and aryl hydrocarbon receptor (AhR) agonistic activities of indole-3-carbinol (I3C) acid condensation products.
I3C is a breakdown product [isothiocyanate] of glucobrassicin. Most biological activities attributed to I3C are believed to result from its acid condensation products, as it is expected that after ingestion of cruciferous vegetables, I3C is completely converted in the stomach before it reaches the intestine.
The reaction mixture was prepared from I3C under acidic conditions. Based on the various HPLC peaks, 9 fractions were collected and tested.
DIM (3,3-diindolylmethane) displayed clear estrogenic activity, showing an additive effect when co-exposed with low concentrations of E2 [estradiol] (below EC50) [effective concentration that gives half-maximal-response of a biological pathway]. However, an anti-estrogenic activity was observed when DIM was co-exposed with higher concentrations of E2, i.e. above EC50. None of the nine fractions was able to inhibit response of E2.
I3C and DIM showed clear anti-androgenic activities when co-exposed with concentrations of T [testosterone] at EC50 or ECmax. DIM showed a relatively strong antagonistic activity, and was able to completely inhibit response of T.
All fractions displayed an AhR agonist activity. Poor activity of fraction 3 seems surprising, as it contains ICZ, which was shown to be a strong AhR agonist. This is a strong indication that ICZ is only present at a very low concentration.
Observed estrogenic and anti-androgenic effects of the reaction mixture are most likely due to DIM.
The present study is the first that demonstrates that DIM also possesses anti-estrogenic properties when co-administered with E2 concentrations above EC50. Rather than ICZ, LTr1 and several other compounds present in fractions 1 and 4 (CTr), and larger molecules present in fractions 7, 8 (LTe1) and 9 seem responsible for observed AhR activity of the reaction mixture.”
https://www.sciencedirect.com/science/article/pii/S1878535220302811 “Acid condensation products of indole-3-carbinol and their in-vitro (anti)estrogenic, (anti)androgenic and aryl hydrocarbon receptor activities”
I came across this study as a result of its citation in Brassica Bioactives Could Ameliorate the Chronic Inflammatory Condition of Endometriosis.
A second 2016 study was with humans:
“Forty-five subjects consumed vegetables, a mixture of brussels sprouts and/or cabbage, at one of seven discrete dose levels of glucobrassicin ranging from 25 to 500 μmol, once daily for 2 consecutive days.
‘Blue Dynasty’ cabbage contained 33.5 ± 4.0 μmol glucobrassicin per 100 grams food weight. ‘Jade Cross’ brussels sprouts contained 206.0 ± 12.9 μmol per 100 grams.
At 50 μmol, variability in 24-hour urinary DIM levels appears to stem from both within an individual and between individuals. At 200 and 500 μmol dose levels, most variability is coming from between individuals rather than within an individual.
Inter-individual DIM variability may reflect the relative benefit an individual derives from consuming glucobrassicin from vegetables, responsive not only to how much glucobrassicin was consumed but also to variations in I3C uptake and DIM metabolism, many of which are not characterized.
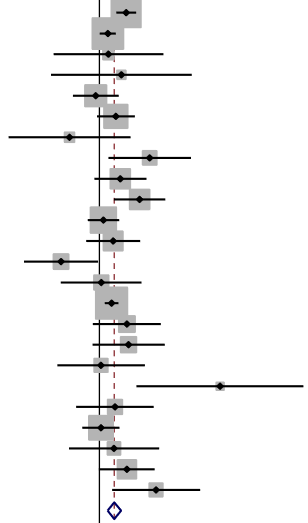
Dose curve between glucobrassicin dose (25–500 μmol) [25, 50, 100, 200, 300, 400, 500] and urinary DIM. Bars represent SD. Estimated parameters in the original scale (95% CI): Maximum DIM 421.5 pmol/mL (154.7–1,148.4), minimum DIM 5.4 pmol/mL (0.7–44.3), EC50 90.2 μmol (29.1–151.3).
We conclude that urinary DIM is a reliable biomarker of glucobrassicin exposure and I3C uptake and that feeding glucobrassicin beyond 200 μmol did not consistently lead to more urinary DIM. Our data support the notion that cancer-preventive properties that might be derived from cruciferous vegetable consumption may require neither a large quantity of vegetables nor high-dose supplements.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5220883/ “Harnessing the Power of Cruciferous Vegetables: Developing a Biomarker for Brassica Vegetable Consumption Using Urinary 3,3′-Diindolylmethane”
1. Most subjects had trouble eating 500 μmol / 242.72 grams of Jade Cross brussels sprouts:
“At the 500 μmol dose level, two subjects could not finish due to the taste of the raw Brussels sprouts and were reassigned to 50 μmol dose level.
Two of the remaining five subjects at the 500 μmol dose level “Did not eat all of the assigned vegetables.” 🙂 That amount of brussels sprouts may have made two more sick because one “Missed one void during 2–6 hour collection period” and another “Missed 2 voids during the 6–12 hour collection period.”
2. From its supplementary material, there were ten subjects who ate a 200 μmol glucobrassicin dose. That’s a lot of raw cabbage (179.10 g) and brussels sprouts (67.96 g).
- On Day 1 at the 2-6 hour point, Subject 27’s urinary DIM measured 10.21 pmol/mL and Subject 20’s measured 991.88, > 9700% higher.
- At that 2-6 hour point on Day 2, the same subjects measured 16.15 and 687.44 pmol/mL, > 4200% higher.
- From Table 1, their respective Mean 24-h DIM ± SE, pmol/mL measurements were 20.7 ± 4.0 and 1105 ± 45, > 5300% higher.
The 100 μmol glucobrassicin dose was 149.25 g Blue Dynasty cabbage and 24.27 g Jade Cross brussels sprouts. Could you eat that every day?
3. There’s sufficient data to make individual DIM bioavailability calculations. Don’t know why this study didn’t do that, nor did any of its 18 citing papers.
One study came close for broccoli and radish sprouts, 2017’s Bioavailability and new biomarkers of cruciferous sprouts consumption (not freely available) by researchers in the same group as Our model clinical trial for Changing to a youthful phenotype with broccoli sprouts. They didn’t disclose and analyze individual DIM bioavailability evidence, though:
“Broccoli and radish sprouts content in GB [glucobrassicin] were ~11.4 and ~7.7 μmol/20 g F.W, respectively. After ingestion of broccoli sprouts, 49% of GB was suitably metabolised and excreted as hydrolysis metabolites, calculated as the sum of I3C and DIM (~5.57 μmol /24 h). Following radish ingestion, the percentage of GB hydrolysed and absorbed was 38% (~2.92 μmol /24 h).
These results of bioavailability contrast with the extremely low percentage (< 1%) of GB excreted as DIM after consumption of Brussels sprouts and cabbage in a previous study (Fujioka, et al., 2014). Further studies about conversion of other indole GLS [glucosinolates] to I3C and DIM are needed to know more about bioavailability of these compounds, as there is no information in literature.”
A ten-subject study in Microwave broccoli seeds to create sulforaphane found inter-individual variability of sulforaphane and its metabolites in blood plasma for the highest and lowest individuals was > 500% (2.032 / 0.359 μmol). The urinary % of dose excreted by the same subjects was > 400% higher (86.9% and 19.5%, respectively.)
These studies present an opportunity for further discovery:
- Which researchers will try to understand causal experiences in people’s lives that produced such effects?
- Which researchers will produce evidence for factors that make people responsively either alive or dead to external influences on their internal environment?
- Where are studies that show when an individual needs to change their responses – their phenotype – they can successfully do so?


 Herding, the story of 2020
Herding, the story of 2020