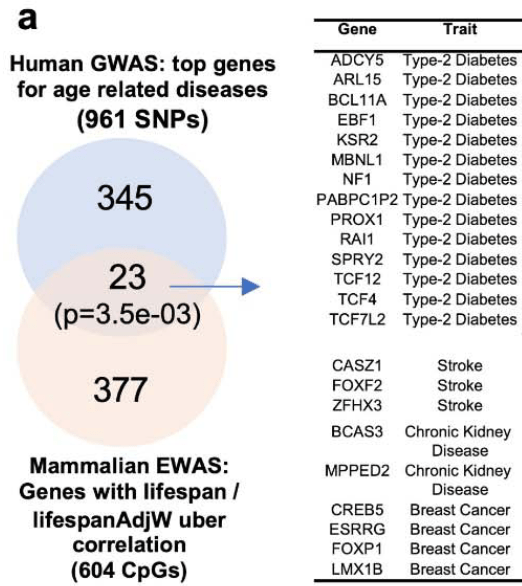
This 2022 rodent and human cell study investigated the smooth muscle cell mineralocorticoid receptor:
“Vascular stiffness increases with age and independently predicts cardiovascular disease risk. Epigenetic changes, including histone modifications, accumulate with age, but the global pattern has not been elucidated nor are the regulators known.
Rising mineralocorticoid receptor (MR) in aging vascular smooth muscle cells downregulates EZH2 to globally shift to a more open chromatin thereby allowing MR to be recruited to promoters to transcriptionally upregulate target genes involved in vascular stiffness. This mechanism provides multiple potential targets to prevent vascular stiffness in aging humans.

We demonstrate for the first time that:
- MR expression increases with age in primary, low passage, human aortic smooth muscle cell (SMC) and correlates with age in whole aortic tissue from aging humans;
- The global proteomic profile of histone modifications in mouse vessels changes profoundly with aging with a significant overall decrease in H3K27 methylation;
- Expression of H3K27 methyltransferase EZH2 decreases with age in mouse vessels and in human SMCs in a MR-dependent manner and negatively correlates with MR expression in whole human aortic tissue;
- The aging-induced decline in EZH2 associates with reduced H3K27 methylation and increased H3K27 acetylation in vitro and in vivo;
- These epigenetic changes in aging human SMC and mouse vessels correspond with increased expression of the vascular stiffness genes, CTGF and integrin-α5, previously identified vascular MR target genes;
- Induction of an aging phenotype in human SMC associates with increased MR enrichment and H3K27 acetylation at these stiffness gene promoters; and
- Inhibition of MR in aged mice and aged human aortic SMCs reverses the entire process; increasing EZH2 and H3K27 methylation, increasing locus-specific EZH2 enrichment and decreasing H3K27 acetylation at stiffness gene promoters, decreasing vascular expression of CTGF and integrin-α5, and decreasing the stiffness and adhesiveness of aged human SMC in vitro and mouse aortic stiffness and fibrosis in vivo.”
https://academic.oup.com/cardiovascres/advance-article-abstract/doi/10.1093/cvr/cvac007/6502304 “Smooth muscle mineralocorticoid receptor as an epigenetic regulator of vascular ageing” (not freely available) Thanks to Dr. Seung Kyum Kim for providing a copy.