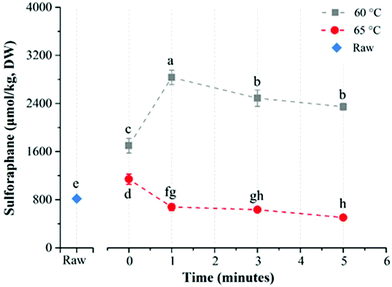
This 2020 Chinese study experimented with enhancing sulforaphane content of broccoli florets in a range of conditions:
“For direct water blanching at 60°C, sulforaphane yield increased with treatment time from 1698.0 ± 121.9 μmol per kg DW (0 min) to 2833.3 ± 118.6 μmol per kg DW (1 min) and then steadily decreased to the lowest value of 2345.8 ± 57.7 μmol per kg DW for 5 min.
The sulforaphane yield of broccoli after 5 min thermal treatment at 65 °C was even lower than the value obtained for raw broccoli. The reason could be leaching of glucoraphanin into blanching water coupled with partial inactivation of myrosinase resulting in low yield of sulforaphane.
For direct water blanching, the best treatment temperature for maximizing sulforaphane yield was 60 °C.
Sulforaphane yield depends on the relative activity of myrosinase and ESP in the broccoli matrix and 3 min treatment at 65 °C during in-pack processing in this study was found to be the best condition that favours conversion into sulforaphane instead of sulforaphane nitrile. This indicates that the condition favours inactivation of ESP to a larger extent while maintaining sufficient myrosinase activity resulting in optimal conversion into sulforaphane.
Under this condition, it seems that all extractable glucoraphanin is converted to sulforaphane assuming 1 to 1 conversion, since glucoraphanin content of broccoli samples were determined to be 3141.2 μmol per kg DW whereas the sulforaphane yield was 3983 μmol per kg DW. The slightly higher sulforaphane yield than would be predicted from the level of glucoraphanin in raw broccoli requires further investigation.”
https://pubs.rsc.org/en/content/articlehtml/2020/fo/c9fo02089f “Mild heat combined with lactic acid fermentation: a novel approach for enhancing sulforaphane yield in broccoli puree”
1. The study presented evidence for kitchen practices:
- Per the above graphic’s point a, I’ve changed to let broccoli sprout heating continue for 1 minute after microwaving to achieve up to but no more than 60°C (140°F). This allows further myrosinase hydrolization of glucoraphanin into sulforaphane. My practice had been to immediately cool them down, which was suboptimal point c on the 60°C line. I still transfer broccoli sprouts to a strainer immediately after microwaving.
- The 60°C cliff finding of Microwave broccoli to increase sulforaphane levels was confirmed by this study’s 65°C direct blanching finding.
2. I didn’t view this study’s in-pack or lactic acid bacteria fermentation findings as having practical kitchen use. Maybe it’s a cultural difference?
3. Poor performance at 65°C after 5 minutes was partially attributed to “leaching of glucoraphanin into the blanching water.” But poor 65°C performance was evident at the 1 minute point compared with good 60°C performance.
“Partial inactivation of myrosinase” at 65°C was more likely to be the dominant factor.
4. Regarding:
“The slightly higher sulforaphane yield than would be predicted from the level of glucoraphanin in raw broccoli requires further investigation.”
The microwaving study author was on a productive investigation track with:
“Microwave irradiation might help to release more conjugated forms of glucosinolates and then get hydrolyzed by released myrosinase.”
That track developed in part from finding that broccoli florets microwaved on full power to 60°C increased glucoraphanin past control (raw) levels:
“The control GLR amount was 2.18 µmol/g DW, while the HL60 GLR amount was 2.78 µmol/g DW.”
Not to mention the coincident 1,114% increase in sulforaphane content of ordinary broccoli purchased at a grocery store!
I arrived at this study through it being referenced in the enjoyable 2020 Spanish review Functional Ingredients From Brassicaceae Species: Overview and Perspectives. Those reviewers noted that this study’s 2019 predecessor Fermentation for enhancing the bioconversion of glucoraphanin into sulforaphane and improve the functional attributes of broccoli puree (not freely available) found:
“Preferential formation of SFN-nitrile (less potential as inducer of phase II detoxification enzymes than SFN) instead of SFN.”