A request was made to present studies that investigated epigenetic impacts of corporal punishments or physical trauma to children or adolescents. Here’s a follow-on of the 2015 Grokking an Adverse Childhood Experiences (ACE) score, since physical abuse is one factor of an ACE score.
1. The largest problem is that a person filling out an ACE questionnaire or Childhood Trauma Questionnaire can’t provide first-hand answers of their own experiences during womb life, infancy, and early childhood. These critical development periods are more impacted by adversity than are later life windows.
Human brains aren’t developed enough before age 3 to provide retrospective answers using cerebral memories. A self-reported ACE score can’t possibly address what happened during the times when we were most vulnerable to disrupted neurodevelopment. And good luck with parents providing factual histories of whether they physically or emotionally neglected, physically or emotionally abused, or otherwise adversely treated their fetus, infant, and young child.
2. Another problem is researchers can pretty much choose whatever questions they want as input criteria. I’ve seen pliable ACE scores developed from 5- to 25-item questionnaires.
Do these questionnaires cover all relevant adverse childhood experiences? For example, are researchers permitted to use as inputs societal-created adversities a child may have lived through such as the Khmer Rouge or Cultural Revolution? Studies are just starting to investigate adverse childhood experiences created by worldwide abuses of authority since 2020.
3. Other problems were discussed in a 2023 paper https://www.sciencedirect.com/science/article/abs/pii/S0145213423003162 “Adverse childhood experiences and adult outcomes using a causal framework perspective: Challenges and opportunities” (not freely available), two of which were:
- Adding up ACE factors to a cumulative score ignores the impact of synergistic sets. For example, although both cumulative ACE scores are 2, a child who was physically and sexually abused would probably be more adversely affected than a child whose parents divorced or separated, and also had a family member incarcerated.
- At any given time point, and especially with older people, there’s a potential selection bias against those most affected by adverse childhood experiences, such as those who died.
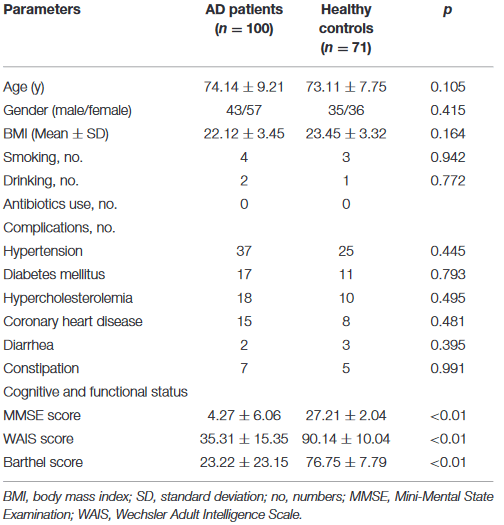
Using flawed, squishy, cumulative ACE scores as inputs, here are two 2023 studies that found epigenetic associations:
“We tested the following pre-registered hypotheses: Mothers’ adverse childhood experiences are correlated with DNA methylation (DNAm) in peripheral blood during pregnancy (hypothesis 1) and in cord blood samples from newborn infants (hypothesis 2), and women’s depression and anxiety symptoms during pregnancy mediate the association between mothers’ ACE exposure and prenatal/neonatal DNA methylation (hypothesis 3).
- Hypothesis 1: In 896 mother−infant pairs with available methylation and ACE exposure data, there were no significant associations between mothers’ ACE score and DNAm from antenatal peripheral blood, after controlling for covariates.
- Hypothesis 2: In infant cord blood, there were 5 CpG sites significantly differentially methylated in relation to mothers’ ACEs (false discovery rate < .05), but only in male offspring. Effect sizes were medium. CpG sites were in genes related to mitochondrial function and neuronal development in the cerebellum.
- Hypothesis 3: There was no mediation by maternal anxiety/depression symptoms found between mothers’ ACEs score and DNAm in the significant CpG sites in male cord blood.”
https://www.jaacap.org/article/S0890-8567(23)00313-1/fulltext “Epigenetic Intergenerational Transmission: Mothers’ Adverse Childhood Experiences and DNA Methylation”
“In this study, the effect of cumulative ACEs experienced on human maternal DNAm was estimated while accounting for interaction with domains of ACEs in prenatal peripheral blood mononuclear cell samples. Intergenerational transmission of ACE-associated DNAm was explored used paired maternal and neonatal cord blood samples. Replication in buccal samples was also explored.
We used a four-level categorical indicator variable for ACEs exposure: none (0 ACEs), low (1–3 ACEs), moderate (4–6 ACEs), and high (> 6 ACEs). 🙄

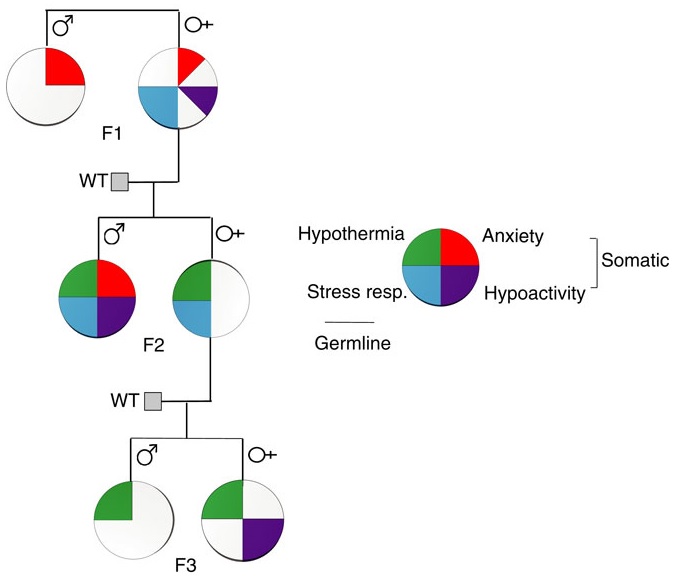
https://www.researchsquare.com/article/rs-2977515/v1 “Effect of Parental Adverse Childhood Experiences on Intergenerational DNA Methylation Signatures”