This 2018 Washington rodent study investigated ovarian disease in F3 great-granddaughters caused by their F0 great-grandmothers’ exposures to DDT or vinclozolin while pregnant:
“Two of the most prevalent ovarian diseases affecting women’s fertility and health are Primary Ovarian Insufficiency (POI) and Polycystic Ovarian Syndrome (PCOS). POI is characterized by a marked reduction in the primordial follicle pool of oocytes and the induction of menopause prior to age 40. POI currently affects approximately 1% of female population. While genetic causes can be ascribed to a minority of patients, around 90% of POI cases are considered idiopathic, with no apparent genetic link nor known cause.
PCOS is a multi-faceted disease that affects 6-18% of women. It is characterized by infrequent ovulation or anovulation, high androgen levels in the blood, and the presence of multiple persistent ovarian cysts.
For both PCOS and POI other underlying causes such as epigenetic transgenerational inheritance of disease susceptibility have seldom been considered. Epigenetic transgenerational inheritance is defined as “the germline transmission of epigenetic information and phenotypic change across generations in the absence of any continued direct environmental exposure or genetic manipulation.” Epigenetic factors include:
The majority of transgenerational studies have examined sperm transmission of epigenetic changes due to limitations in oocyte numbers for efficient analysis.
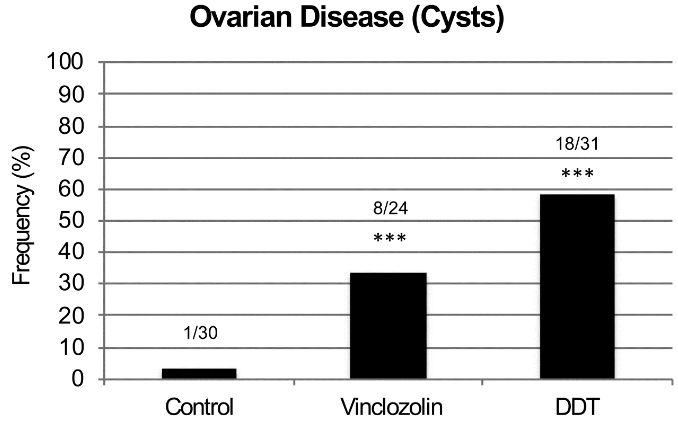
There was no increase in ovarian disease in direct fetal exposed F1 [grandmothers] or germline exposed F2 [mothers] generation vinclozolin or DDT lineage rats compared to controls.
The transgenerational molecular mechanism is distinct and involves the germline (sperm or egg) having an altered epigenome that following fertilization may modify the embryonic stem cells epigenome and transcriptome. This subsequently impacts the epigenetics and transcriptome of all somatic cell types derived from these stem cells.
Therefore, all somatic cells in the transgenerational [F3] animal have altered epigenomes and transcriptomes and those sensitive to this alteration will be susceptible to develop disease. The F3 generation can have disease while the F1 and F2 generations do not, due to this difference in the molecular mechanisms involved.
The epimutations and gene expression differences observed are present in granulosa cells in the late pubertal female rats at 22-24 days of age, which is long before any visible signs of ovarian disease are detectable. This indicates that the underlying factors that can contribute to adult-onset diseases like PCOS and POI appear to be present early in life.
Ancestral exposure to toxicants is a risk factor that must be considered in the molecular etiology of ovarian disease.”
1. The study highlighted a great opportunity for researchers of any disease that frequently has an “idiopathic” diagnosis. It said a lot about research priorities that “around 90% of POI cases are considered idiopathic, with no apparent genetic link nor known cause.”
It isn’t sufficiently explanatory for physicians to continue using categorization terminology from thousands of years ago. Science has progressed enough with measured evidence to discard the “idiopathic” category and express probabilistic understanding of causes.
2. One of this study’s coauthors made a point worth repeating in The imperative of human transgenerational studies: What’s keeping researchers from making a significant difference in their fields with human epigenetic transgenerational inheritance studies?
3. Parts of the study’s Discussion section weren’t supported by its evidence. The study didn’t demonstrate:
- That “all somatic cells in the transgenerational animal have altered epigenomes and transcriptomes”; and
- The precise “molecular mechanisms involved” that exactly explain why “the F3 generation can have disease while the F1 and F2 generations do not.”
https://www.tandfonline.com/doi/abs/10.1080/15592294.2018.1521223 “Environmental Toxicant Induced Epigenetic Transgenerational Inheritance of Ovarian Pathology and Granulosa Cell Epigenome and Transcriptome Alterations: Ancestral Origins of Polycystic Ovarian Syndrome and Primary Ovarian Insuf[f]iency” (not freely available)