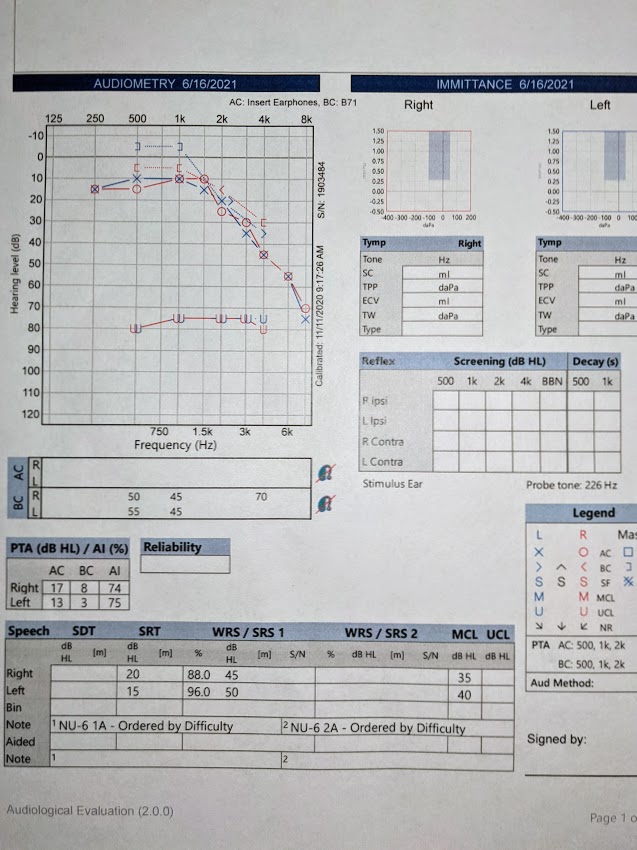
This 2020 review focused on three Vitamin K-dependent proteins (VKDPs):
“We summarize three important emerging VKDPs: Growth arrest‑specific protein 6 (Gas 6), Gla‑rich protein (GRP) and periostin in terms of their functions in physiological and pathological conditions. As examples:
- Carboxylated Gas 6 and GRP effectively protect blood vessels from calcification;
- Gas 6 protects from acute kidney injury and is involved in chronic kidney disease;
- GRP contributes to bone homeostasis and delays progression of osteoarthritis; and
- Periostin is involved in all phases of fracture healing and assists myocardial regeneration in the early stages of myocardial infarction.
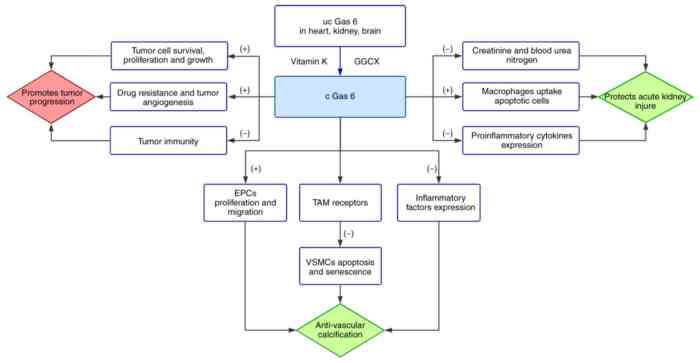
The ‘+’ refers to promotion and ‘-‘ refers to inhibition. Green represents Gas 6 physiological effects and red represents its pathological effects.
- Gas 6 resists vascular calcification: i) Gas 6 promotes proliferation and migration of endothelial progenitor cells (EPCs); ii) Gas 6 inhibits apoptosis and senescence of vascular smooth muscle cells (VSMCs) by binding Tyro3, Axl and Mer (TAM) receptors; iii) Gas 6 decreases expression of inflammatory factors, including TNF-α and ICAM-1.
- Gas 6 protects from acute kidney injury: i) Gas 6 significantly reduces creatinine and blood urea nitrogen; ii) Gas 6 enhances macrophages to uptake apoptotic cells; iii) Gas 6 reduces the expression of pro-inflammatory cytokines, such as IL-1β.
- Gas 6 assists tumor progression: i) Gas 6 is necessary for survival, proliferation and growth of tumor cells; ii) Gas 6 contributes to drug resistance and tumor angiogenesis; iii) Gas 6 negatively regulates tumor immunity.
Numerous physiological benefits of vitamin K2 have been identified, such as anti-vascular calcification, glycemic control, and lipid-lowering effects. However, some questions about relationships between vitamin K2 and cancers remain unsolved. VKDPs are expected to be biomarkers for many diseases.”
https://www.spandidos-publications.com/10.3892/ijmm.2020.4835?text=fulltext “Role of emerging vitamin K‑dependent proteins: Growth arrest‑specific protein 6, Gla‑rich protein and periostin (Review)”
This review’s VKPD biomarkers included:
- Vascular calcification;
- Asthma;
- Bronchial obstruction;
- Diabetic nephropathy; and
- Fracture risk.
Elaborating on this last item:
“In a cohort of 607 postmenopausal women from France that were followed up for 7 years, a positive correlation between serum periostin and fracture risk was observed. The association was independent of bone mineral density and prior fractures, indicating that periostin is an independent predictive marker of fracture risk.”
As pointed out in Chronological age by itself is an outdated clinical measurement, bone mineral density is one of several historical measurements that were selected for their relative convenience instead of chosen for their efficacy. We’re in a different century now.