A trio of papers, with the first a 2017 review exploring broccoli sprout compounds’ effects on head hair:
“Skin appendages, notably hair follicles (HFs), can be exposed to high levels of reactive oxygen species (ROS), which are generated through metabolic reactions occurring mostly in the mitochondria, peroxisomes, and endoplasmic reticulum, as well as in the plasma membrane. Despite their involvement in redox stress and cellular damage, ROS also have key roles in physiological signalling processes, including but not limited to, control of stem cell quiescence / differentiation, regulation of innate and adaptive immune responses and importantly, normal HF development.
HFs are composed of a series of concentric keratinocyte layers with a central hair shaft, all of which are encapsulated by a mesenchymal connective tissue sheath. Within this structure is an area known as the ‘bulge’, housing a population of epithelial and melanocyte stem cells. The hair bulb, the lowermost portion of the HF, contains transient amplifying cells that produce rapidly proliferating matrix keratinocytes that give rise to various cell types of the inner root sheath and hair shaft itself.
Putative impact of NRF2 activation on protection against hair disorders:
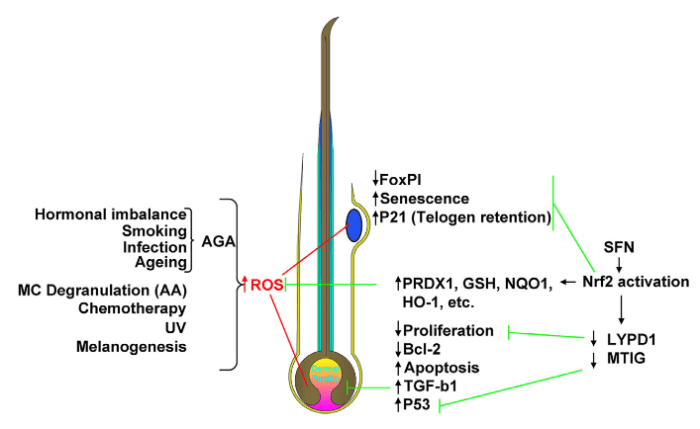
- Accumulation of excess ROS within crucial HF compartments (i.e. bulb and bulge) can be induced by endogenous and exogenous stressors associated with androgenetic alopecia (AGA), alopecia areata (excessive mast cell degranulation), chemotherapy, UV exposure, and even physiological processes such as melanogenesis.
- In the HFSCs [hair follicle stem cells] of the bulge, this can lead to reduced FOXP1 signaling, increased senescence and P21-mediated telogen retention, contributing to hair ageing.
- In the hair bulb, negative consequences of excessive ROS can include reduced matrix keratinocyte proliferation and Bcl-2 expression, coupled to increased p53 activity and apoptosis. This redox imbalance may also stimulate dermal papilla-derived TGF-b1 release associated with AGA.
- NRF2 activation via SFN [sulforaphane] can induce expression of numerous downstream targets, hence suggesting the potential to counteract excessive ROS and associated pathologies, for example via enhanced clearance of reactive species, detoxification, NADPH generation, and GSH maintenance.
- In addition, NRF2 may down-regulate genes that would negatively impact on proliferation and stimulate apoptosis.
- Ultimately, activation of NRF2 has the potential to protect against HF miniaturization, chemotherapy-induced apoptosis, HFSC aging, and hair greying, through maintenance of normal redox homeostasis.
Whereas eumelanin (black) is involved in natural UV protection by reducing generation of free radicals, pheomelanin (red) can trigger generation of ROS. It would certainly be interesting to determine whether NRF2 activity is therefore higher in individuals with red as opposed to black hair, in order to mitigate any negative impact from higher ROS generation.
Modulation of NRF2 activity is an attractive approach for further study in prevention of hair greying and HFSC ageing. The remarkable prospect for NRF2 activators in modulating other oxidative stress-linked disease states, strongly advocates for development of NRF2 targeting as a novel strategy in modulating redox-associated disorders of the HF.”
https://onlinelibrary.wiley.com/doi/abs/10.1002/bies.201700029 “Oxidative stress management in the hair follicle: Could targeting NRF2 counter age‐related hair disorders and beyond?” (not freely available)
This review was cited in a 2020 Exploring the possibility of predicting human head hair greying from DNA using whole-exome and targeted NGS data study:
“This study aimed to assess the potential of genetic data to predict hair greying in a population of nearly 1000 individuals from Poland. Most prediction information was brought by age alone. Genetic variants explained < 10% of hair greying variation, and the impact of particular SNPs on prediction accuracy was found to be small.
Study population included 673 males (67.4%) and 325 (32.6%) females. Mean age of participants was 30.5 ± 8.8.
Hair greying was recorded in 14.3% of individuals aged 18–30, and prevalence of grey hair was noted to be significantly higher in young males when comparing to young females (17.8 and 9.2%, respectively). Incidence of grey hair increased to 29.5% in the group of people aged 18–40 years, and was 84.2% when people aged ≥40 years were considered.
Because pleiotropy is so common, it would be impossible to predict natural phenotypes avoiding genes involved in determination of pathological phenotypes. The penetrance of individual SNP variants is usually low, and they altogether can only explain a small fraction of predisposition to disease.
Prediction of hair greying status solely based on genetic information is currently impossible.”
A 2020 review had a pertinent evaluation scheme:
“Geroprotectors are pharmacological agents that decrease the rate of aging and extend lifespan. We proposed a set of primary and secondary selection criteria for potential geroprotectors. Primary criteria:
- Life extension in experiments with wild type animal models. The geroprotector should prolong life of the model beyond the intact maximum lifespan, protecting it from one or more mechanisms of aging.
- Improvement of molecular, cellular, and physiological biomarkers to a younger state, or slow down progression of age-related changes in humans.
- Most potential geroprotectors are preventive only when applied at relatively high concentrations. The lifespan-extending dose should be several orders of magnitude less than the toxic dose.
- Minimal side effects at the therapeutic dosage at chronic application.
- The potential benefit of taking a geroprotector may come after a long period. Potential geroprotectors should initially improve some parameters of health-related quality of life: physical, mental, emotional, or social functioning of the person.”
https://www.mdpi.com/2076-3921/9/6/529/htm “Terpenoids as Potential Geroprotectors”