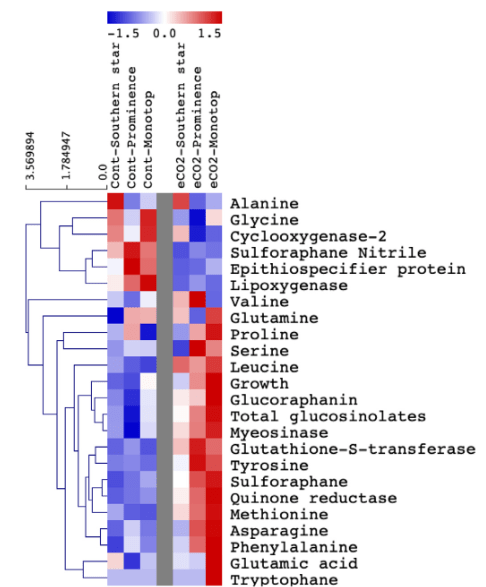
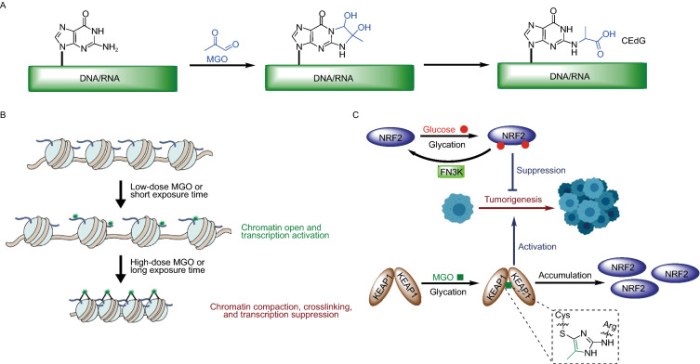
This 2020 paper reviewed hormetic effects of a broccoli sprout compound:
“Sulforaphane (SFN) induces a broad spectrum of chemoprotective effects across multiple organs that are of importance to public health and clinical medicine. This chemoprotection is dominated by hormetic dose responses that are mediated by the Nrf2/ARE pathway and its complex regulatory interactions with other factors and pathways, such as p53 and NF-κB.
The stimulatory zone for in vitro studies proved to be consistently in the 1-10 μM range. Hormetic studies of SFN strongly targeted activation of Nrf2.
Capacity to activate Nrf2 diminishes with age, and may affect capacity of SFN to effectively enhance adaptive responses.
A 4-hour exposure induced a 24 hour Nrf2-mediated increase in enzymes that reduce free-radical damage in neurons and astrocytes. Repeated 4-hour treatment for four days affected an accumulation along with a persistent protection.
In the case of continuous exposure to SFN, such as taking a daily supplement, SFN treatment did not result in an accumulation of HMOX1 [heme oxygenase (decycling) 1 gene] mRNA or protein. This suggested that HMOX1 response may experience feedback regulation, avoiding possible harmful overproduction.”
https://www.sciencedirect.com/science/article/abs/pii/S1043661820315917 “The phytoprotective agent sulforaphane prevents inflammatory degenerative diseases and age-related pathologies via Nrf2-mediated hormesis” (not freely available)
One coauthor has been on a crusade to persuade everybody of this paradigm. Hormesis’ hypothesis isn’t falsifiable in all circumstances, however.
Hormetic effects may be experimental considerations. But what’s the point of performing sulforaphane dose-response experiments in contexts that are physiologically unachievable with humans? Two examples:
- Autism biomarkers and sulforaphane:
“There was no concentration-dependence in induction of any genes examined, with the higher (5 μM) concentration of SF even showing a slightly diminished effect for induction of AKR1C1 and NQO1. Although this concentration is achievable in vivo, more typical peak concentrations of SF (and its metabolites) in human plasma are 1-2 μM.”
- Human relevance of rodent sulforaphane studies:
“Over two-thirds of the animal studies have used doses that exceed the highest (and bordering on intolerable) doses of sulforaphane used in humans. The greater than 4-log spread of doses used in mice appears to be driven by needs for effect reporting in publications rather than optimization of translational science.”
This paper cited many hormetic effects that were human-irrelevant without making a distinction. But it also had parts such as:
“The capacity for high concentrations of AITC [allyl isothiocyanate] to enhance genetic damage is not relevant since such high concentrations are not realistically achievable in normal human activities.
Humans ingest only the R-isomer of SFN via diet. Their dosing strategy adopted concentrations of R-SFN that were less than those employed to induce cytotoxic effects in cancer cells and that simulated its consumption as a dietary supplement.”

 Landing eagle
Landing eagle
 Young dolphins eating breakfast
Young dolphins eating breakfast





 Flying over waves
Flying over waves



 Diving for breakfast
Diving for breakfast Owl before sunrise
Owl before sunrise