The founder of the epigenetic clock methodology with the coauthor of Aging as an unintended consequence released a 2020 rodent study “Reversing age: dual species measurement of epigenetic age with a single clock” at https://www.biorxiv.org/content/10.1101/2020.05.07.082917v1.full.pdf:
“We employed six clocks to investigate the rejuvenation effects of a plasma fraction treatment in different rat tissues. Two of these epigenetic clocks apply to both humans and rats.
The treatment more than halved the epigenetic ages of blood, heart, and liver tissue. A less pronounced, but statistically significant, rejuvenation effect could be observed in the hypothalamus.
The treatment was accompanied by progressive improvement in the function of these organs as ascertained through numerous biochemical/physiological biomarkers and behavioral responses to assess cognitive functions. Cellular senescence, which is not associated with epigenetic aging, was also considerably reduced in vital organs.
Plasma fraction treatment consists of two series of intravenous injections of plasma fraction. Rats were injected four times on alternate days for 8 days. A second identical series of injections were administered 95 days later. In its entirety, the experiment lasted 155 days.
Overall, this study demonstrates that a plasma-derived treatment markedly reverses aging according to epigenetic clocks and benchmark biomarkers of aging.”
The study hasn’t been peer reviewed, so can’t be viewed yet as conclusive. Given that researchers’ single-most valuable asset is their reputations, though, will the findings have major revisions?
I was alerted to the study by Josh Mitteldorf’s blog post Age Reduction Breakthrough, who did his usual excellent curation:
“Most of the explosion in aging research (and virtually all the venture capital startups) are looking to treat aging at the cellular level. Their paradigm is that aging is an accumulation of molecular damage, and they see their job as engineering of appropriate repair mechanisms.
The truth, as Katcher [the lead lab researcher] understands it, is that, to a large extent, aging is coordinated system-wide via signal molecules in the blood. The problem is that there are thousands of constituents represented in tiny concentrations in blood plasma, but conveying messages that cells read. Which of these are responsible for aging?
The two-species clock[s] was [were] a significant innovation, a first bridge for translating results from an animal model into their probable equivalent in humans. Besides the methylation clock[s], the paper presents evidence of rejuvenation by many other measures. For example:
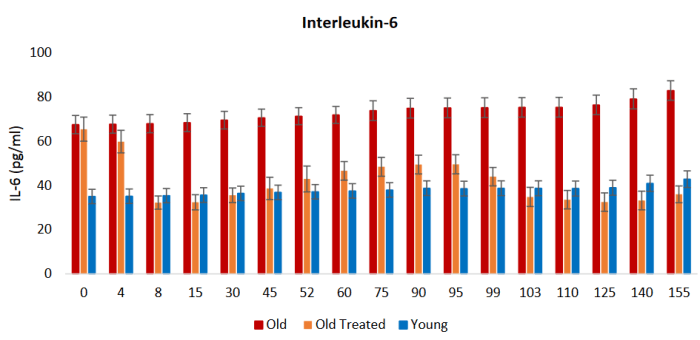
- IL-6, a marker of inflammation, was restored to low youthful levels;
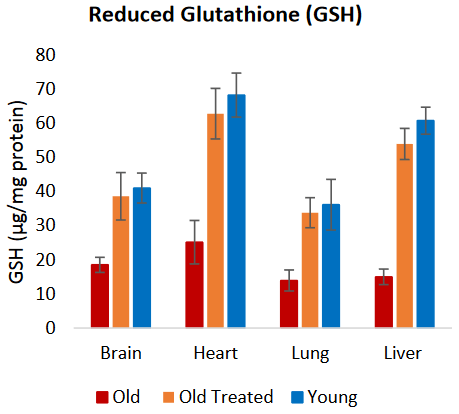
- Glutathione (GSH), superoxide dismutase (SOD), and other antioxidants were restored to higher youthful levels;
- In tests of cognitive function (Barnes maze), treated rats scored better than old rats, but not as well as young rats.;
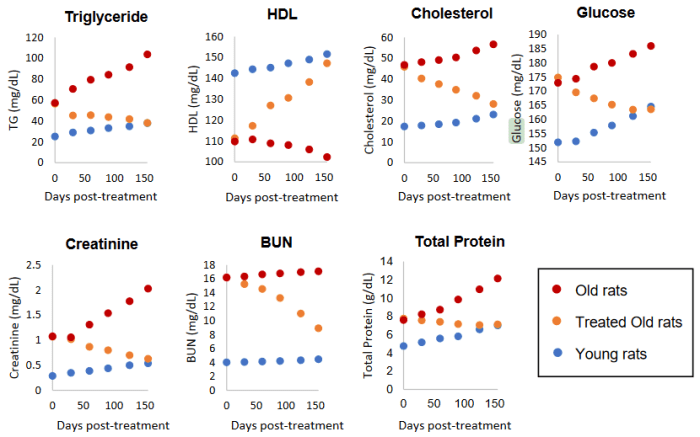
- Blood triglycerides were brought down to youthful levels;
- HDL cholesterol rose to youthful levels; and
- Blood glucose fell toward youthful levels.
These results bring together three threads that have been gaining credibility over the last decade. Mutually reinforcing, the three have a strength that none of them could offer separately.
- The root cause of aging is epigenetic progression = changes in gene expression over a lifetime.
- Methylation patterns in nuclear DNA are not merely a marker of aging, but its primary source. Thus aging can be reversed by reprogramming DNA methylation.
- Information about the body’s age state is transmitted system-wide via signal molecules in the blood. Locally, tissues respond to these signals and adopt a young or an old cellular phenotype as they are directed.”
Several of these aging measurements are also positively affected by sulforaphane. Using Sulforaphane: Its “Coming of Age” as a Clinically Relevant Nutraceutical in the Prevention and Treatment of Chronic Disease as a reference:
1. “Chronic inflammation”
“Antioxidants in general and glutathione in particular can be depleted rapidly under conditions of oxidative stress, and this can signal inflammatory pathways associated with NF-κB. SFN [sulforaphane] has been shown to inhibit NF-κB in endothelial cells.
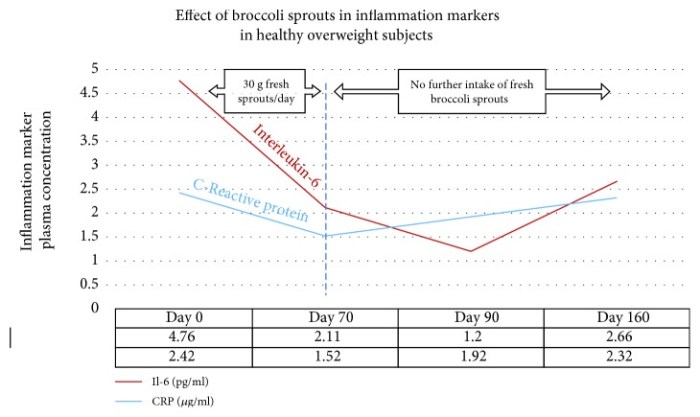
Two key inflammatory cytokines were measured at four time points in forty healthy overweight people [our model clinical trial, Effects of long-term consumption of broccoli sprouts on inflammatory markers in overweight subjects]. The levels of both interleukin-6 (Il-6) and C-reactive protein (CRP) declined over the 70 days during which the sprouts were ingested. These biomarkers were measured again at day 90, wherein it was found that Il-6 continued to decline, whereas CRP climbed again. When the final measurement was taken at day 160, CRP, although climbing, had not returned to its baseline value. Il-6 remained significantly below the baseline level at day 160.”
2. “Oxidative stress”
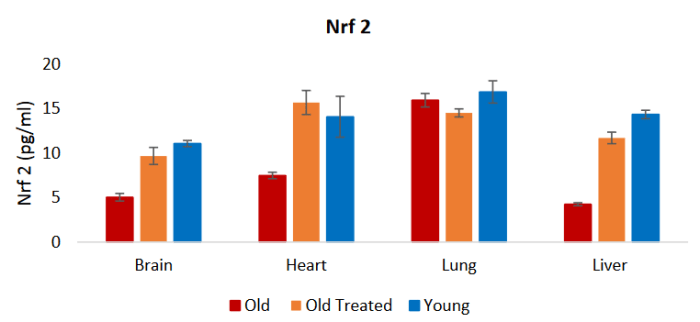
“As a mediator for amplification of the mammalian defence system against various stressors, Nrf2 [nuclear factor erythroid 2-related factor 2] sits at the interface between our prior understanding of oxidative stress and the endogenous mechanisms cells use to deal with it. Diseases known to be underpinned by oxidative stress are proving to be more responsive to amplification of cellular defences via Nrf2 activation than by administration of direct-acting antioxidant supplements.
SFN, with absolute bioavailability of around 80%, [is] capable of increasing several endogenous antioxidant compounds via the transcription factor, Nrf2.
Nrf2 is ubiquitously expressed with the highest concentrations (in descending order) in the kidney, muscle, lung, heart, liver, and brain. Nrf2 was shown to prevent endothelial cells from exhibiting a proinflammatory state. Nrf2 is required for protection against glucose-induced oxidative stress and cardiomyopathy in the heart.
Well in excess of 500 genes have been identified as being activated by SFN via the Nrf2/ARE [Antioxidant Response Element] pathway, and it is likely that this underestimates the number as others are being discovered. Of the available SFN clinical trials associated with genes induced via Nrf2 activation, many demonstrate a linear dose-response. More recently, it has become apparent that SFN can behave hormetically with different effects responsive to different doses.
It [sulforaphane] is not only a potent Nrf2 inducer but also highly bioavailable so that modest practical doses can produce significant clinical responses. Other Nrf2 activators [shown in the above image] not only lack potency but also lack the bioavailability to be considered as significant intracellular Nrf2 activators.”
The study’s most relentlessly questioned, scrutinized, and criticized findings may be the two new epigenetic clocks that apply to both humans and rats. The researchers invited other researchers to validate these clocks because:
“If validated, this would be a step change in aging research. Although conservation of aging mechanism could be equally deduced from the existence of multiple individual clocks for other mammals (mouse, dog), the single formula of the human-rat clock that is equally applicable to both species effectively demonstrates this fact.”
The commonalities of this study with efforts to change my inflammatory phenotype with broccoli sprouts were summarized in the Discussion section:
“Apart from rejuvenating the vital organs of the treated rats, plasma fraction also impacted two fundamental physiological processes that underlie a great number of pathologies, namely oxidative stress and inflammation. Within a week of treatment, the markers of chronic inflammation (IL-6 and TNF-α) were significantly reduced and remained low throughout the entire experiment.
Likewise, markers of oxidative stress in brain, heart, lung and liver, which were very much higher in control old rats, were at the end of the experimental period, indistinguishable between plasma fraction-treated old rats and young ones. Concomitant with this drastic reduction in oxidative stress was the augmented levels of antioxidants (GSH, Catalase and SOD) in these tissues, indicating that modulating the levels of ROS [reactive oxygen species] to that of youthful rats is at least one way by which plasma fraction suppresses oxidative stress. It remains to be ascertained whether the rate of ROS generation is also reduced.
The levels of Nrf2, a transcription factor that impacts on oxidative stress, as well as inflammation, were raised by plasma fraction treatment of old rats to those of the young ones, indicating yet another level by which this treatment modulates these two critical processes. Collectively, these results show that plasma fraction treatment impacts not only the overt performances of organs, but also the underlying physiological processes that are pivotal for optimal organ function and health.”
Great stuff, huh? Are you ready to change your phenotype?
Continued with Part 2 of Rejuvenation therapy and sulforaphane.