This 2020 Swiss review subject was the interaction of Nrf2 activators and skin:
“The electrophile and Nrf2 activator dimethyl fumarate (DMF) is an established and efficient drug for patients suffering from the common inflammatory skin disease psoriasis. DMF is being tested for pharmacological activity in several other inflammatory skin conditions.

Psoriasis is a chronic inflammatory skin disease affecting 2–4% of the population and plaque psoriasis is the most common type, affecting about 90% of all patients. As about 30% of all patients suffer from moderate and severe psoriasis, there is a strong need for efficient systemic treatment options with few side effects.
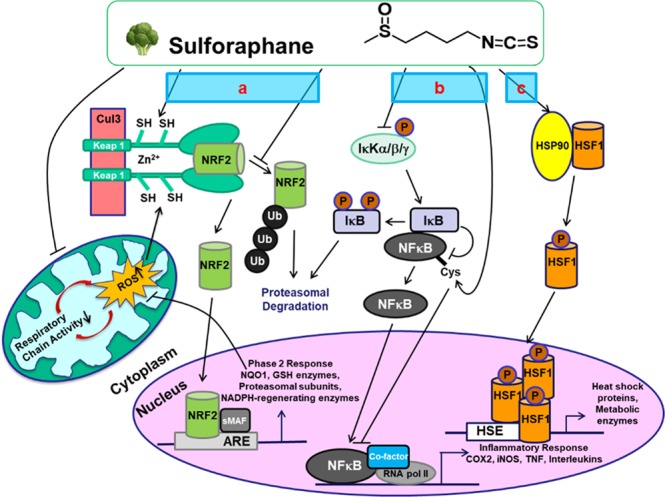
SFN [sulforaphane] blocks NF-κB activity by several mechanisms. SFN oxidizes IκB, thereby inhibiting its phosphorylation and downstream NF-κB activation, but also targets specific cysteine residues of p50/p65, causing a reduction in DNA binding.
More indirect effects have also been suggested. SFN induces HO-1 expression via Nrf2, which in turn inhibits NF-κB. The isothiocyanate can also react with and oxidize components of cellular redox buffers, such as glutathione and thioredoxin, which are required to retain NF-κB’s DNA-binding capacity.
NLRP3 is believed to be critically involved in common diseases, whereas its role in immunity is rather minor. The mechanisms underlying NLRP3 inflammasome activation are of high medical interest.
Electrophiles can directly inhibit inflammasome activation. SFN inhibits activation of NLRP3 in the absence of NRF2 expression in a very fast manner, suggesting that transcriptional effects are not relevant for NLRP3 inhibition. SFN inhibits NLRP3 even in KEAP1 knockout cells.
All these results demonstrate that electrophiles can inhibit the inflammasome pathway in a direct manner, perhaps via the modification of reactive cysteine residues of inflammasome proteins or those which regulate activation of these complexes.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7072181/ “Electrophiles against (Skin) Diseases: More Than Nrf2”
These reviewers focused on a pharmaceutical. Read this article for progress made on a generic:
“Biogen is expected to appeal this ruling against its Tecfidera patent protection, which is not due to expire until 2028. Its list price is reported to be about $2,026 for a two-week (14 day) supply at 120 mg.
‘The District Court decision clears the legal pathway for us to bring our dimethyl fumarate product to market, and we are working with the FDA to accelerate our regulatory approval target action date, which currently is November 16.'”
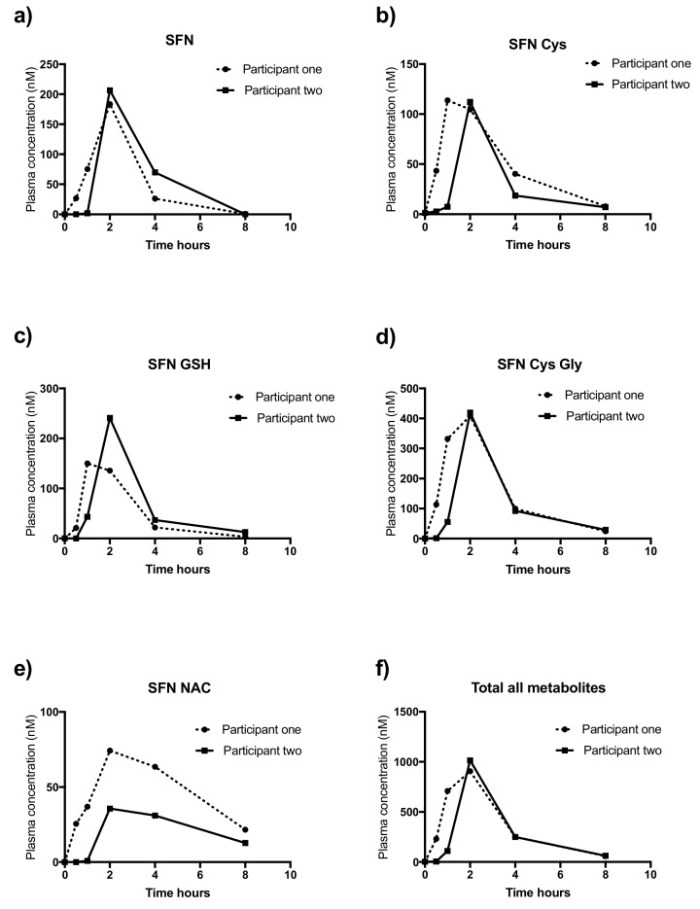
Broccoli or Sulforaphane: Is It the Source or Dose That Matters? listed 15 mouse studies and 4 human studies of sulforaphane treatments of skin diseases in Supplementary Material Table S3.
From Novel Nrf2 activators from microbial transformation products inhibit blood–retinal barrier permeability in rabbits:
“The cysteine residue of sulforaphane works as a weak electrophile and it interacts with cysteine residues of Keap1. Dimethyl fumarate is also a weak electrophile.
Nrf2 activity was evaluated by NQO1 induction activity in Hepa1c1c7 cells. RS9 was the most potent and the concentration needed to double (CD) the specific Nrf2 activity was 0.2 nM. The CD values for bardoxolone methyl, sulforaphane and dimethyl fumarate were 0.9 nM, 154.4 nM and 13.3 μM respectively.”

1. This review didn’t mention dimethyl fumarate’s NQO1 induction CD value because..? It’s one of Nrf2 signaling pathway’s main studied parameters, along with HO-1. For example, from Autism biomarkers and sulforaphane:
“This time point was chosen based on our earlier observations of kinetics of upregulation of Nrf2-dependent genes by SF, and was expected to capture increased mRNA production of both very fast (HO-1) and relatively slow (NQO1) responders.”
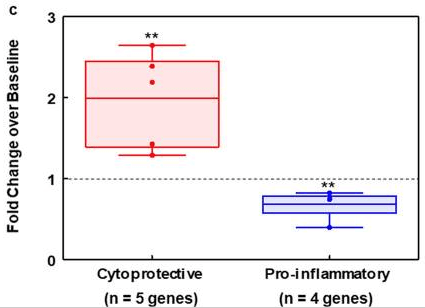
2. What about adverse effects? From Sulforaphane and RNAs:
“DMF is the most successful Nrf2 activator, FDA-approved in 2013 for the treatment of relapsing remitting multiple sclerosis. However, DMF causes leukopenia and other side-effects.
Bardoxolone cleared Phase II clinical trials for the treatment of advanced chronic kidney disease and type 2 diabetes mellitus, but was halted in Phase III trials due to cardiovascular concerns.”
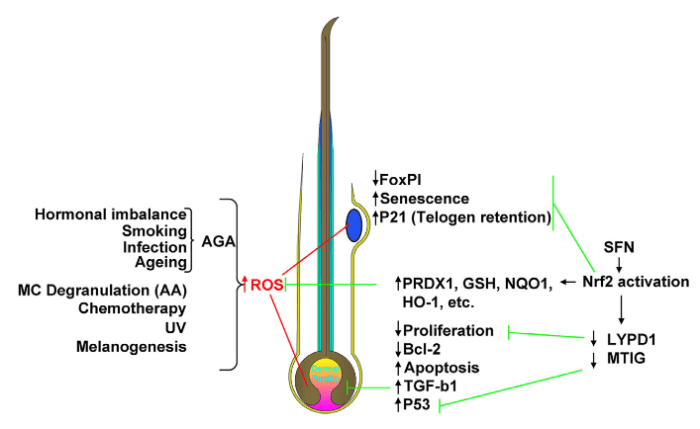
3. What about prevention mechanisms for skin problems? Skin care isn’t just cancer prevention.
So – what can a person do to treat an inflammation problem in our largest organ, our skin?
- Pay $2,026 every two weeks to take a daily 120 mg dose of a brand name pharmaceutical?
- Wait around for some hypothetical future “development of new tailor-made molecules and drugs for the many inflammatory conditions which are associated with Nrf2, NF-κB and inflammasomes”?
- Try other treatments that just address symptoms, not causes?
Or eat daily clinically-relevant broccoli sprout dosages for < $500 a year?