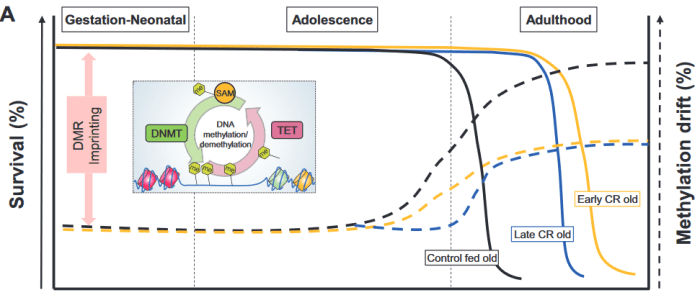
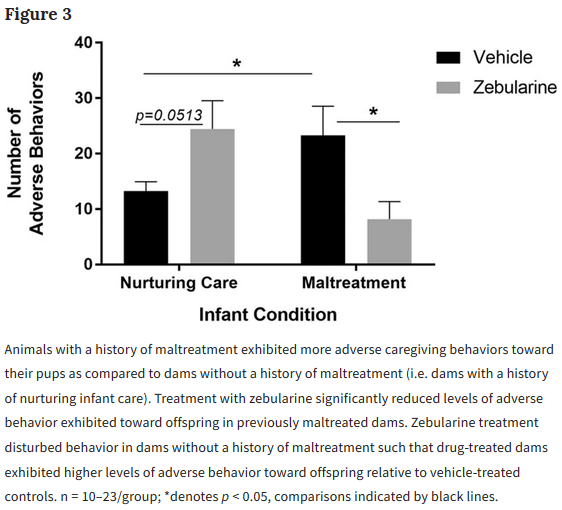
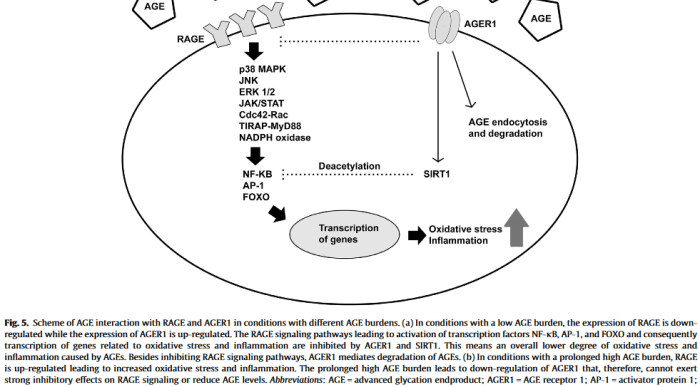
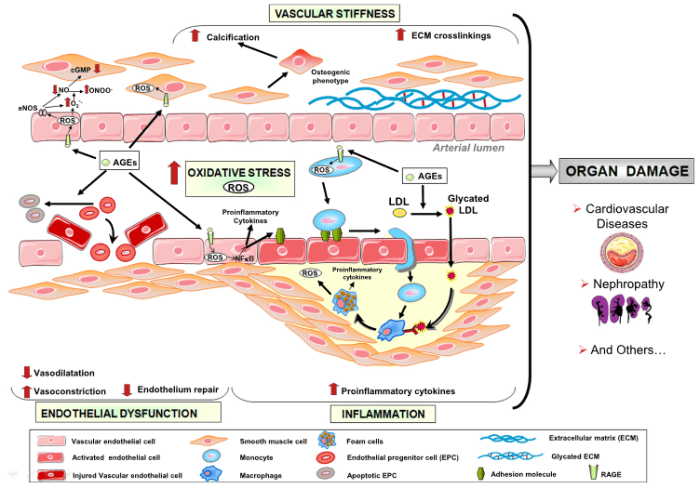
This 2019 Germany/Canada human fetal cell study was a Proceedings of the National Academy of Sciences of the United States of America direct submission:
“In a human hippocampal progenitor cell line, we assessed the short- and long-term effects of GC [glucocorticoid] exposure during neurogenesis on messenger RNA expression and DNA methylation profiles. Our data suggest that early exposure to GCs can change the set point of future transcriptional responses to stress by inducing lasting DNAm changes.”
The study’s basic finding was that cells had initial responses to stressors that primed them for subsequent stressors. Since this finding wasn’t new, the researchers tried to make it exciting by applying it to novel contexts that were yet circumscribed by official paradigms.
Hypothesis-seeking associations of human fetal hippocampal cell behaviors with human behaviors were flimsy stretches, as were correlations to placental measurements. These appeared to have been efforts to find headline-making effects.
There wasn’t even a hint of the principle described in Epigenetic variations in metabolism:
“Because of the extreme interconnectivity of cell regulatory networks, even at the cellular level, predicting the impact of a sequence variant is difficult as the resultant variation acts:
- In the context of all other variants and
- Their potential additive, synergistic and antagonistic interactions.
This phenomenon is known as epistasis.”
It would have condemned pet models of reality to admit that a cell exists in multiple contexts of other cells with potential additive, synergistic, and antagonistic interactions.
A research proposal to trace a specific cell type’s behaviors – while isolated from their extremely interconnected networks – to trillion-celled human behaviors would be rejected in less-politicized organizations.
Sanctioned speculations manifested in this paper with phrases such as “although not significant..” and “although not directly tested..” The study’s title was probably a disappointment in that it conformed to the study’s evidence.
Involvements of psychiatry departments at the pictured Kings College, Harvard, etc., as part of PNAS entrenched politics, retard advancements of science past approved paradigms.
This is my final curation of PNAS papers.
https://www.pnas.org/content/pnas/early/2019/08/08/1820842116.full.pdf “Glucocorticoid exposure during hippocampal neurogenesis primes future stress response by inducing changes in DNA methylation”