This 2019 US review subject was caloric restriction (CR) without malnutrition:
“Cellular adaptation that occurs in response to dietary patterns can be explained by alterations in epigenetic mechanisms such as DNA methylation, histone modifications, and microRNA. Epigenetic reprogramming of the underlying chronic low-grade inflammation by CR can lead to immuno-metabolic adaptations that enhance quality of life, extend lifespan, and delay chronic disease onset.
Short- and long-term CRs produce significant changes in different tissues and across species, in some animal models even with sex-specific effects. Early CR onset may cause a different and even an opposite effect on physiological outcomes in animal models such as body weight.”
https://academic.oup.com/advances/article-abstract/10/3/520/5420411 “Epigenetic Regulation of Metabolism and Inflammation by Calorie Restriction” (not freely available)
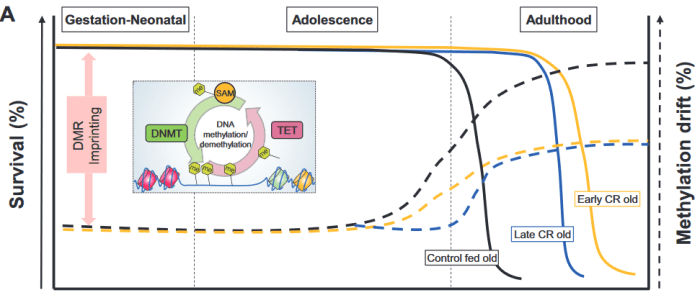
1. The review didn’t present evidence to equate survival (left axis) with methylation drift (right axis) per the above graphic. Methylation drift should point in the opposite direction of survival, if anything.
2. No mention was made of the epigenetic clock method of measuring age acceleration, although it’s been available since 2013 and recent diet studies have used it. The sole citation of an age acceleration study was from 2001, which was unacceptable for a review published in 2019.
3. The review provided many cellular-level details about the subject. However, organism-level areas weren’t sufficiently evidenced:
A. Arguments for an effect usually include explanations for no effect as well as for opposite effects. The reviewers didn’t provide direct evidence for why, if caloric restriction extended lifespan, caloric overabundance produced shorter lifespans.
B. Caloric restriction evidence was presented as if only it was responsible for organism-level effects. Other mechanisms may have been involved.
An example of such a mechanism was demonstrated in a 2007 rodent study Reduced Oxidant Stress and Extended Lifespan in Mice Exposed to a Low Glycotoxin Diet which compared two 40%-calorie-restricted diets.
The calories and composition of both diets were identical. However, advanced glycation end product (AGE) levels were doubled in standard chow because heating temperatures were “sufficiently high to inadvertently cause standard mouse chow to be rich in oxidant AGEs.”
The study found that a diet with lower chow heating temperatures increased lifespan and health span irrespective of caloric restriction!
- The low-AGE calorie-restricted diet group lived an average of 15% longer (>20 human equivalent years) than the CR group.
- 40% of the low-AGE calorie-restricted diet group were still alive when the last CR group member died.
- The CR group also had significantly more: 1) oxidative stress damage; 2) glucose and insulin metabolism problems; and 3) kidney, spleen, and liver injuries.