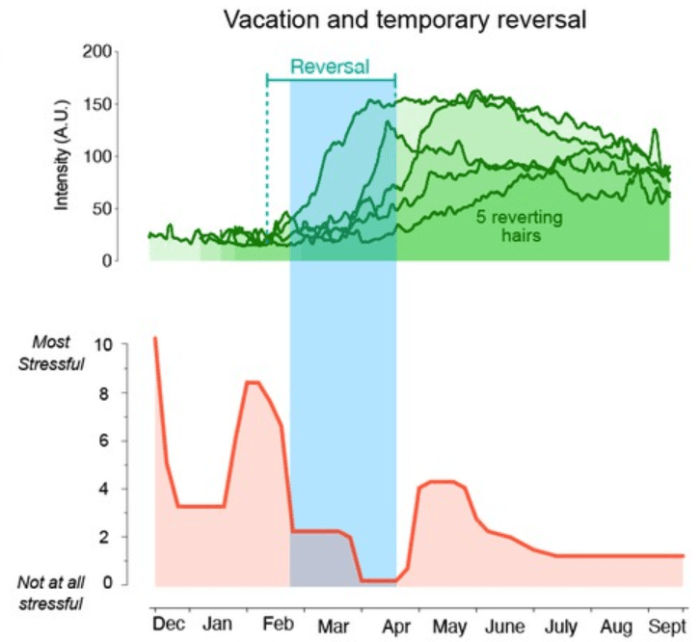
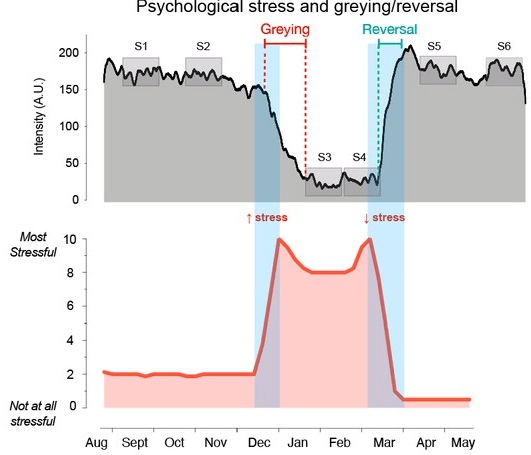
Two papers on aging measurements, starting with a 2022 human study:
“We collected longitudinally across the adult age range a comprehensive list of phenotypes within four domains (body composition, energetics, homeostatic mechanisms and neurodegeneration / neuroplasticity) and functional outcomes. We integrated individual deviations from population trajectories into a global longitudinal phenotypic metric of aging.

We demonstrate that accelerated longitudinal phenotypic aging is associated with faster physical and cognitive decline, faster accumulation of multimorbidity, and shorter survival.”
https://www.nature.com/articles/s43587-022-00243-7 “Longitudinal phenotypic aging metrics in the Baltimore Longitudinal Study of Aging”
I disagree with this study’s methodology.
1. Although it acknowledged individual variability, nothing was done to positively adjust to those facts. What could have been done per A review of biological variability was:
“Obtain a measurement of variability that is independent of the mean to ensure to not confound changes in variability with shifts in mean.”
2. A usual research practice is to take at least three measurements, and use their average as representative. That wasn’t done here, maybe because of time and expense considerations?
3. An important measurement for physical function was the time to finish a 400 meter walk. I walk more than ten times that almost every day. I use the first 400 meters as a warmup period while getting to the beach to walk eastward and enjoy the predawn light and water animal activity. I concentrate on gait speed during the last third while walking westward on a straightaway bike path.
This study would measure my gait speed as a sometimes old person during the first 400 meters, rather than a gait speed that usually approaches a young person’s during the last 400 meters. Even if I tried to walk my fastest right out of the gate, I wouldn’t be surprised to find a decade or two difference by this study’s measurements between a morning walk’s first and last 400 meter gait speeds.
4. An important cognitive function measurement was the Digital Symbol Substitution Test, apparently taken during subjects’ fasted state? Sometimes after exercising, I’m okay cognitively when starting work in a fasted state at 6:30 a.m., and other times I’m tired.
Two days ago during the last hour of work 1:30-2:30 p.m., I did outstanding work, four hours after eating whole oats for breakfast, and after drinking two coffees and three teas. I took time to put together pieces of puzzles into proper contexts for management’s attention. My bosses weren’t too pleased with the story it told, but it is what it is.
5. Are measurements of how you start what matters? Or is it how you finish, as is common in competitive sports?
This study would measure my cognitive function as a sometimes old person, rather than performance that approaches a young person’s later in the workday. For both physical and cognitive function, my abilities to ramp up and come close to young people’s capabilities are features that I work on, not random, inconvenient measurement variability.
6. Blood measurements were downgraded as having “limited coverage of the four phenotypic domains.” These were taken to fit into specific paradigms and epigenetic clocks. They predictably failed to show causality, as acknowledged with:
“Our analysis showed strong associations between global longitudinal phenotypic score and changes in physical and cognitive function. We did not have sufficient observations to fully separate these two dimensions over time, which would have strengthened the assumption of causality.”
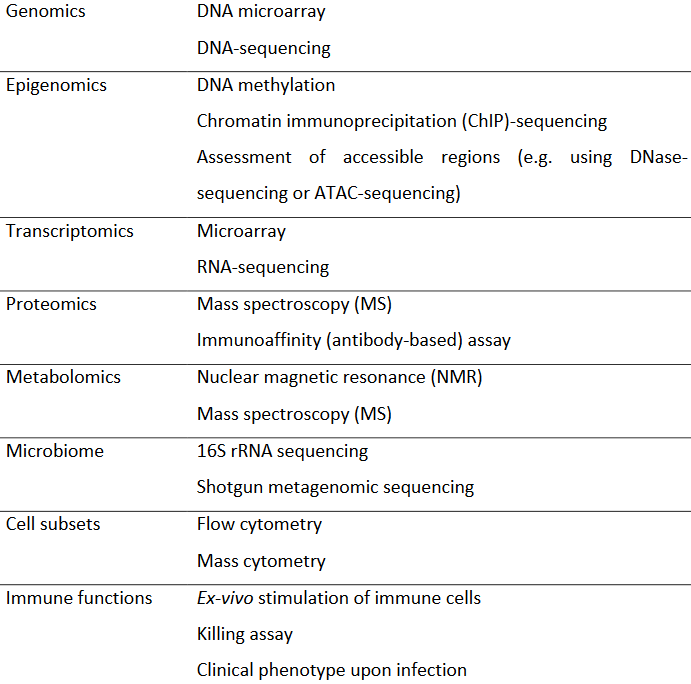
Nowhere in this study was it hinted that all measurements were downstream effects of unmeasured causes. A follow-on study could reanalyze these subjects’ blood samples, MRI, and other measurements for originating upstream factors of signaling pathways and cascades per Signaling pathways and aging and An environmental signaling paradigm of aging.
Reference 35 of this first study was a 2021 human and rodent study that was tossed in as a limitation with:
“We might not have all of the relevant phenotypic measures (for example, more detailed immune profiles) for all participants.”
Its findings included:
“From the blood immunome of 1,001 individuals aged 8–96 years, we developed a deep-learning method based on patterns of systemic age-related inflammation. The resulting inflammatory clock of aging (iAge) tracked with multimorbidity, immunosenescence, frailty and cardiovascular aging, and is also associated with exceptional longevity in centenarians.
Canonical markers of acute infection such as IL-6 and tumor necrosis factor-α were not major contributors to iAge, indicating that, except for IL-1β, infection-driven inflammatory markers of the acute inflammatory response do not contribute to age-related chronic inflammation.
We conducted a follow-up study in an independent cohort of 97 extremely healthy adults (aged 25–90 years) matched for cardiovascular risk factors (including conserved levels of high-sensitivity C-reactive protein), selected from a total of 151 recruited participants using strict selection criteria. In this healthy cohort, inflammation markers were measured using a 48-plex cytokine panel. Only 6 circulating immune proteins were significantly correlated with age, with CXCL9 again the largest contributor to age-related inflammation.
CXCL9 is a T-cell chemoattractant induced by IFN-γ and is mostly produced by neutrophils, macrophages and endothelial cells (ECs). We find that CXCL9 is mainly produced by aged endothelium and predicts subclinical levels of cardiovascular aging in nominally healthy individuals.
We did not find any significant correlation between known disease risk factors reported in the study (BMI, smoking, dyslipidemia) and levels of CXCL9 gene or protein expression. We hypothesize that one root cause of CXCL9 overproduction is cellular aging per se, which can trigger metabolic dysfunction.
As ECs but not cardiomyocytes expressed the CXCL9 receptor, CXCR3, we hypothesize that this chemokine acts both in a paracrine fashion (when it is produced by macrophages to attract T cells to the site of injury) and in an autocrine fashion (when it is produced by the endothelium) creating a positive feedback loop. In this model, increasing doses of CXCL9 and expression of its receptor in these cells leads to cumulative deterioration of endothelial function in aging.
IFN-γ did not increase in expression in our cellular aging RNA-seq experiment, suggesting that there are triggers of CXCL9 (other than IFN-γ) that play a role in cellular senescence in the endothelium that are currently unknown. However, in our 1KIP study, IFN-γ was in fact the second-most important negative contributor to iAge, which could be explained by the cell-priming effect of cytokines, where the effect of a first cytokine alters the response to a different one.
iAge derived from immunological cytokines gives us an insight into the salient cytokines that are related to aging and disease. A notable difference compared to other clocks is that iAge is clearly actionable as shown by our experiments in CXCL9 where we can reverse aging phenotypes. More practical approaches range from altering a person’s exposomes (lifestyle) and/or the use of interventions to target CXCL9 and other biomarkers described here.
Our immune metric for human health can identify within healthy older adults with no clinical or laboratory evidence of cardiovascular disease, those at risk for early cardiovascular aging. We demonstrate that CXCL9 is a master regulator of vascular function and cellular senescence, which indicates that therapies targeting CXCL9 could be used to prevent age-related deterioration of the vascular system and other physiological systems as well.”
https://www.nature.com/articles/s43587-021-00082-y “An inflammatory aging clock (iAge) based on deep learning tracks multimorbidity, immunosenescence, frailty and cardiovascular aging”