This 2019 US rodent study concerned transmitting poor maternal care to the next generation:
“The quality of parental care received during development profoundly influences an individual’s phenotype, including that of maternal behavior. Infant experiences with a caregiver have lifelong behavioral consequences.
Maternal behavior is a complex behavior requiring the recruitment of multiple brain regions including the nucleus accumbens, bed nucleus of the stria terminalis, ventral tegmental area, prefrontal cortex, amygdala, and medial preoptic area. Dysregulation within this circuitry can lead to altered or impaired maternal responsiveness.
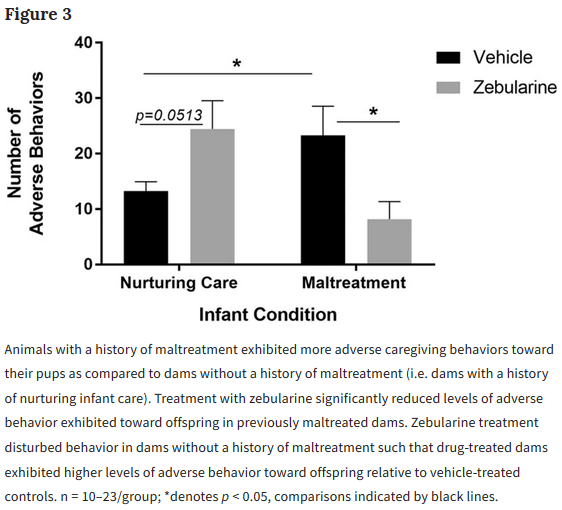
We administered zebularine, a drug known to alter DNA methylation, to dams exposed during infancy to the scarcity-adversity model of low nesting resources, and then characterized the quality of their care towards their offspring.
- We replicate that dams with a history of maltreatment mistreat their own offspring.
- We show that maltreated-dams treated with zebularine exhibit lower levels of adverse care toward their offspring.
- We show that administration of zebularine in control dams (history of nurturing care) enhances levels of adverse care.
- We show altered methylation and gene expression in maltreated dams normalized by zebularine.
These findings lend support to the hypothesis that epigenetic alterations resulting from maltreatment causally relate to behavioral outcomes.
Maternal behavior is an intergenerational behavior. It is important to establish the neurobiological underpinnings of aberrant maternal behavior and explore treatments that can improve maternal behavior to prevent the perpetuation of poor maternal care across generations.”
The study authors demonstrated intergenerational epigenetic effects, and missed an opportunity to also investigate transgenerational epigenetically inherited effects. They cited reference 60 for the first part of the above quotation, but the cited reviewer misused the transgenerational term by applying it to grand-offspring instead of the great-grand-offspring.
There were resources available to replicate the study authors’ previous findings, which didn’t show anything new. Why not use such resources to uncover evidence even more applicable to humans by extending experiments to great-grand-offspring that would have no potential germline exposure to the initial damaging cause?
Could a study design similar to A limited study of parental transmission of anxiety/stress-reactive traits have been integrated? That study’s thorough removal of parental behavior would be an outstanding methodology to confirm by falsifiability whether parental behavior is both an intergenerational and a transgenerational epigenetic inheritance mechanism.
Rodent great-grand-offspring can be studied in < 9 months. It takes > 50 years for human studies to reach the great-grand-offspring transgenerational generation.
- Why not attempt to “prevent the perpetuation of poor maternal care across generations?”
- Isn’t it a plausible hypothesis that humans “with a history of maltreatment mistreat their own offspring?”
- Isn’t it worth the extra effort to extend animal research to investigate this unfortunate chain?
https://www.nature.com/articles/s41598-019-46539-4 “Pharmacological manipulation of DNA methylation normalizes maternal behavior, DNA methylation, and gene expression in dams with a history of maltreatment”