This 2020 review covered interactions of gut microbiota, intestinal mucus, and dietary fibers. I’ve outlined its headings and subheadings, and ended with its overview:
“I. Dietary fibers and human mucus-associated polysaccharides: can we make an analogy?
I.1 Brief overview of dietary fibers and mucus polysaccharides structures and properties
I.I.1 Dietary fibers
- Dietary fiber intake and health effects
I.I.2 Intestinal mucus polysaccharides
I.2 Similarities and differences between dietary fibers and mucus carbohydrates
- Origin and metabolism
- Structure
II. Interactions of dietary fibers and mucus-associated polysaccharides with human gut microbiota
II-1 Substrate accessibility and microbial niches
- Dietary fibers
- Mucus polysaccharides
II-2 Recognition and binding strategies
- Dietary fibers
- Mucus polysaccharides
II-3 Carbohydrate metabolism by human gut microbiota
II-3.1 Specialized carbohydrate-active enzymes
II-3.2 Vertical ecological relationships in carbohydrate degradation
- Dietary fibers
- Mucus polysaccharides
II-3.3 Horizontal ecological relationships in carbohydrate degradation
II.4 Effect of carbohydrates on gut microbiota composition and sources of variability
II.4.1 Well-known effect of dietary fibers on the gut microbiota
II.4.2 First evidences of a link between mucus polysaccharides and gut microbiota composition
III. Gut microbiota, dietary fibers and intestinal mucus: from health to diseases?
[no III.1]
III.2 Current evidences for the relationship between dietary fibers, mucus and intestinal-inflammatory related disorder
III.2.1 Obesity and metabolic-related disorders
- Dietary fibers
- Mucus polysaccharides
III.2.2 Inflammatory bowel diseases
- Dietary fibers
- Mucus polysaccharides
III.2.3 Colorectal cancer
- Dietary fibers
- Mucus polysaccharides
IV. How enteric pathogens can interact with mucus and dietary fibers in a complex microbial background?
IV.1 Mucus-associated polysaccharides: from interactions with enteric pathogens to a cue for their virulence?
IV.1.1 Pathogens binding to mucus
- Binding structures
- Sources of variations
IV.1.2 Mucus degradation by pathogens
- Bacterial mucinases
- Glycosyl hydrolases
IV.1.3 Mucus-based feeding of pathogens
- Primary degraders or cross-feeding strategies
- Importance of microbial background
IV.1.4 Pathogens and inflammation in a mucus-altered context
IV.1.5 Modulation of virulence genes by mucus degradation products
IV.2 How can dietary fiber modulate enteric pathogen virulence?
IV.2.1 Direct antagonistic effect of dietary fibers on pathogens
- Bacteriostatic effect
- Inhibition of cell adhesion
- Inhibition of toxin binding and activity
IV.2.2 Indirect effect of dietary fibers through gut microbiota modulation
- Modulation of microbiota composition
- Modulation of gut microbiota activity
IV.2.3 Inhibition of pathogen interactions with mucus: a new mode of dietary fibers action?
- Binding to mucus: dietary fibers acting as a decoy
- Inhibition of mucus degradation by dietary fibers
V. Human in vitro gut models to decipher the role of dietary fibers and mucus in enteric infections: interest and limitations?
V.1 Main scientific challenges to be addressed
V.2 In vitro human gut models as a relevant alternative to in vivo studies
V.3 In vitro gut models to decipher key roles of digestive secretions, mucus and gut microbiota
V.4 Toward an integration of host responses
V.5 From health to disease conditions

Overview of the potential role of dietary fibers in preventing enteric infections. Reliable and converging data from scientific literature are represented with numbers in circles, while data more hypothetical needing further investigations are represented with numbers in squares.
- Some dietary fibers exhibit direct bacteriostatic effects against pathogens.
- Dietary fiber degradation leads to short-chain fatty acids (SCFAs) production that can modulate pathogens’ virulence.
- By presenting structure similarities with receptors, some dietary fibers can prevent pathogen adhesin binding to their receptors.
- By the same competition mechanism, dietary fibers can also prevent toxins binding to their receptors.
- Dietary fibers are able to promote gut microbiota diversity.
- Dietary fibers may promote growth of specific strains with probiotic properties and therefore exhibit anti-infectious properties.
- Suitable dietary fiber intake prevents microbiota’s switch to mucus consumption, limiting subsequent commensal microbiota encroachment and associated intestinal inflammation.
- Dietary fibers may prevent pathogen cross-feeding on mucus by limiting mucus degradation and/or by preserving diversity of competing bacterial species.
- By preventing mucus over-degradation by switcher microbes, dietary fibers can hamper pathogen progression close to the epithelial brush border, and further restrict subsequent inflammation.”
https://doi.org/10.1093/femsre/fuaa052 “Tripartite relationship between gut microbiota, intestinal mucus and dietary fibers: towards preventive strategies against enteric infections” (not freely available)
There were many links among gut microbiota studies previously curated. For example, Go with the Alzheimer’s Disease evidence found:
“Akkermansia cannot always be considered a potentially beneficial bacterium. It might be harmful for the gut–brain axis in the context of AD development in the elderly.”
The current review provided possible explanations:
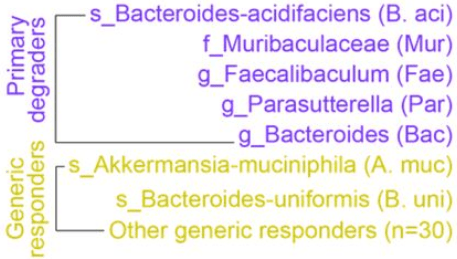
“Akkermansia muciniphila could be considered as a species that fulfills a keystone function in mucin degradation. It is a good example of a mucus specialist.”
Points #7-9 of the above overview inferred that insufficient dietary fiber may disproportionately increase abundance of this species. But Gut microbiota strains also found that effects may be found only below species at species’ strain levels.
These reviewers provided copies in places other than what’s linked above. Feel free to contact them for a copy.
Moon bandit