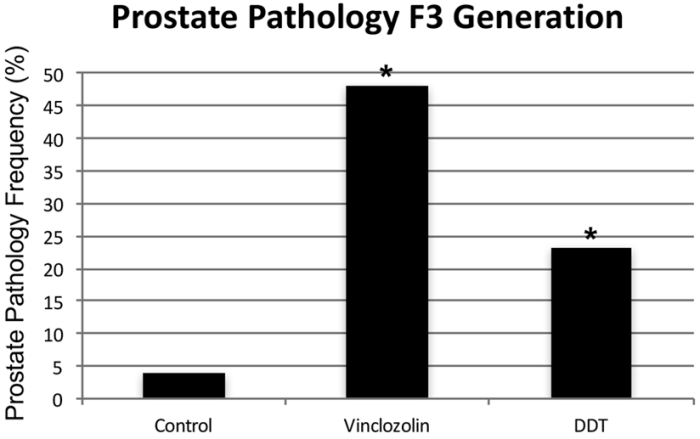
This 2019 Washington State University rodent study from Dr. Michael Skinner’s lab found:
“A cascade of epigenetic alterations initiated in PGCs [primordial germ cells of F3 males] appears to be required to alter epigenetic programming during spermatogenesis to modify the sperm epigenome involved in transgenerational epigenetic inheritance phenomenon.
Following fertilization there is a DNA methylation erasure to generate stem cells in the early embryo, which then remethylate in a cell type-specific manner. DNA methylation erasure is thought to, in part, reset deleterious epigenetics in the germline. However, imprinted gene DNA methylation sites and induced transgenerational epimutations appear to be protected from this DNA methylation erasure.
A germline with an altered epigenome has the capacity to alter the early embryo’s stem cell’s epigenome and transcriptome that can subsequently impact epigenomes and transcriptomes of all derived somatic cells. Therefore, an altered sperm epigenome has the capacity to transmit phenotypes transgenerationally. Experiments have demonstrated that epigenetic inheritance can also be transmitted through the female germline.
Previously, agricultural fungicide vinclozolin was found to promote transgenerational inheritance of sperm differential DNA methylation regions (DMRs) termed epimutations that help mediate this epigenetic inheritance. The current study was designed to investigate developmental origins of transgenerational DMRs during gametogenesis.
The current study with vinclozolin-induced transgenerational inheritance demonstrates that sperm DMRs also originate during both spermatogenesis and earlier stages of germline development, but at distinct developmental stages. Fetal exposure initiates a developmental cascade (i.e., distinct developmental origins) of aberrant epigenetic programming, and does not simply induce a specific number of DMRs that are maintained throughout development.”
https://www.tandfonline.com/doi/pdf/10.1080/15592294.2019.1614417?needAccess=true “Transgenerational sperm DNA methylation epimutation developmental origins following ancestral vinclozolin exposure”
The study’s main hypotheses were:
“Following fertilization, the hypothesis is that transgenerational epimutations modify early embryonic transcriptomes and epigenomes to re-establish the cascade for the next generation.
As the individual develops, all somatic cells have altered epigenomes and transcriptomes to promote disease susceptibility later in life.”
Researchers: adopt these hypotheses, and apply them to human studies.
1. Don’t get off track by requiring that the same phenotype must be observed in each generation for there to be transgenerational epigenetic inheritance, because:
“Fetal exposure..does not simply induce a specific number of DMRs that are maintained throughout development.”
Animal transgenerational studies have shown that epigenetic inheritance mechanisms may both express different phenotypes for each generation, and entirely skip a phenotype in one or more generations!
2. Don’t limit your study designs to F1 children as did:
- DNA methylation and childhood adversity and
- Epigenetics research that was designed to fall one step short of wonderful.
3. Don’t stop at F2 grandchildren as did:
- Burying human transgenerational epigenetic evidence;
- The Not-Invented-Here syndrome; and
- “Transgenerationally” inherited epigenetic effects of fetal alcohol exposure.
4. Continue studies on to F3 great-grandchildren who had no direct exposure to altering stimulus. Keep in the forefront of your research proposals that there are probably more than 10,000,000 F3 descendants of DES-exposed women just in the US!