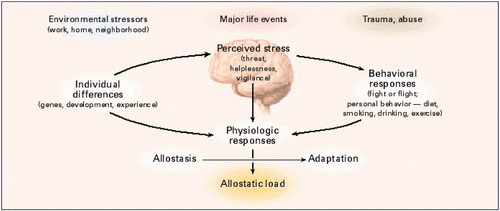
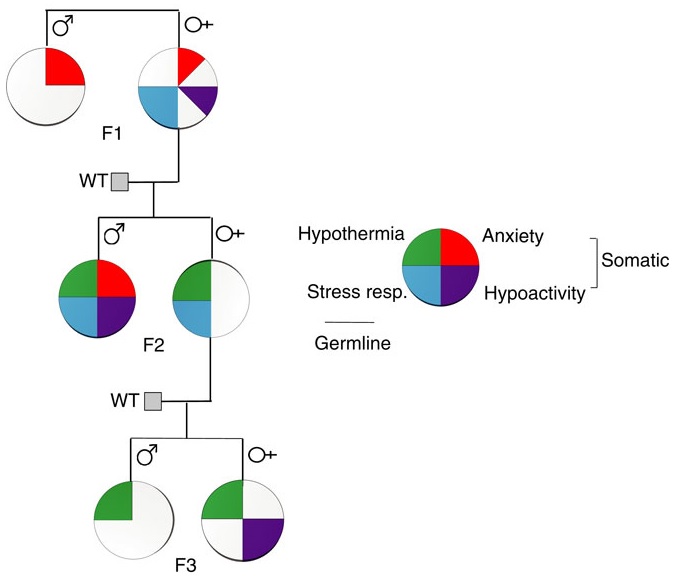
The first paper of Transgenerational epigenetic inheritance week was a 2017 Canadian/Netherlands review that’s organized as follows:
“First, we address mechanisms of developmental and transgenerational programming of disease and inheritance. Second, we discuss experimental and clinical findings linking early environmental determinants to adverse aging trajectories in association with possible parental contributions and sex-specific effects. Third, we outline the main mechanisms of age-related functional decline and suggest potential interventions to reverse negative effects of transgenerational programming.”
A transgenerational phenotype was defined as an epigenetic modification that was maintained at least either to F2 grandchildren in the paternal lineage, or to F3 great-grandchildren in the maternal lineage.
The reviewers noted that mechanisms of transgenerational programming are complex and multivariate. Severity, timing, and type of exposure; lineage of transmission; germ cell exposure; and gender of an organism were the main factors that may determine consequences. Mechanisms reviewed were:
- Parental exposure to an adverse environment;
- Altered maternal behavior and care of offspring; and
- Experience-dependent modifications of the epigenome.
There was a long list of diseases and impaired functionalities that were consequences of ancestral experiences and exposures. Most studies were of animals, but a few were human, such as those done on effects of extended power outages during a Quebec ice storm of January 1998.
One intervention that was effective in reversing a transgenerational phenotype induced by deficient rodent maternal care was to place pups with a caring foster female soon after birth. It’s probably unacceptable in human societies to preemptively recognize all poor-care human mothers and remove the infant to caring foster mothers. But researchers could probably find enough instances to develop studies of the effectiveness of such placements in reversing a transgenerational phenotype.
The review didn’t have suggestions for reversing human transgenerational phenotypes, just “potential interventions to reverse negative effects of transgenerational programming.” Interventions suggested for humans – exercise, enriched lifestyle, cognitive training, dietary regimens, and expressive art and writing therapies – only reduced impacts of transgenerational epigenetic effects.
Tricky wording of “reverse negative effects of transgenerational programming” showed that research paradigms weren’t aimed at resolving causes. The review was insufficient for the same reasons mentioned in How one person’s paradigms regarding stress and epigenetics impedes relevant research, prompting my same comment:
Aren’t people interested in human treatments of originating causes so that their various symptoms don’t keep bubbling up? Why wouldn’t research paradigms be aligned accordingly?
When reversals of human phenotypes aren’t researched, problems may compound by being transmitted to the next generations.
http://www.sciencedirect.com/science/article/pii/S014976341630714X “Transgenerational effects of early environmental insults on aging and disease incidence” (not freely available)