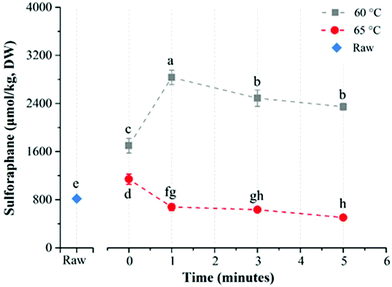
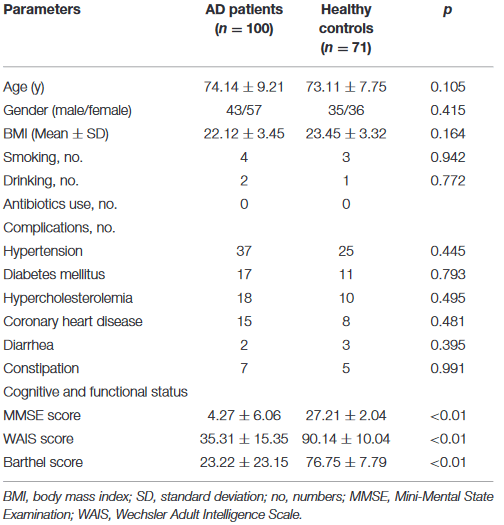
This 2021 study investigated gut microbiota differences between 100 AD patients and 71 age- and gender-matched controls:
“Structural changes in fecal microbiota were evident in Chinese AD patients, with decreased alpha-diversity indices and altered beta-diversity ones, evidence of structurally dysbiotic AD microbiota.
Interestingly, traditionally beneficial bacteria, such as Bifidobacterium and Akkermansia, increase in these AD patients while Faecalibacterium and Roseburia decrease significantly. Different species of Bifidobacterium may have different effects that can explain why Bifidobacterium spp. are commonly associated with healthy and diverse microbiota but sometimes also isolated in other conditions. We needed to re-examine the therapeutic potential of Bifidobacterium in terms of maintaining cognitive function and treating dementia.
Surprisingly, our data indicate that Akkermansia was among the most abundant genera in AD-associated fecal microbiota. Similar to Bifidobacterium, Akkermansia was negatively correlated with clinical indicators of AD, such as MMSE, WAIS, and Barthel, and anti-inflammatory cytokines such as IFN-γ.
Based on our present observations, Akkermansia cannot always be considered a potentially beneficial bacterium. It might be harmful for the gut–brain axis in the context of AD development in the elderly.
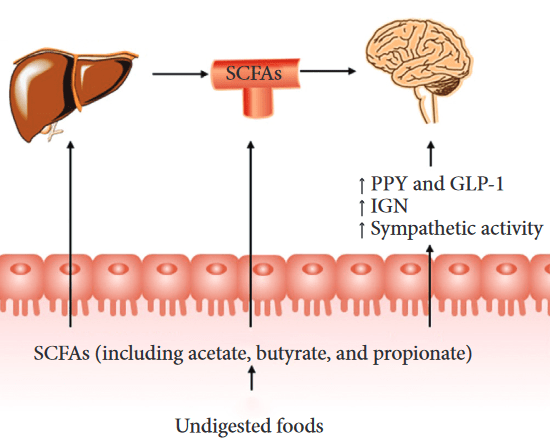
Aging is associated with an over-stimulation of both innate and adaptive immune systems, resulting in a low-grade, chronic state of inflammation defined as inflammaging. This can increase gut permeability and bacterial translocation.
Characteristics of AD microbial profiles changed from butyrate producers, such as Faecalibacterium, into lactate producers, such as Bifidobacterium. These alterations contributed to shifts in metabolic pathways from butyrate to lactate, which might have participated in pathogenesis of AD. Specific roles of AD-associated signatures and their functions should be explored in further studies.”
https://www.frontiersin.org/articles/10.3389/fcell.2020.634069/full “Structural and Functional Dysbiosis of Fecal Microbiota in Chinese Patients With Alzheimer’s Disease”
The control group’s 73-year-olds were better off than AD patients. How were they compared with their previous life stages?
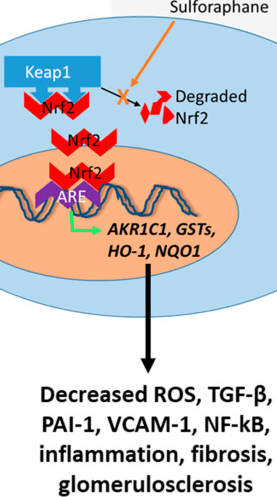
Since we’re all aging, how do we each prepare ourselves? I’ll return to evidence including 2020 A rejuvenation therapy and sulforaphane, recently amplified in Part 2 of Switch on your Nrf2 signaling pathway:
“A link between inflammation and aging is the finding that inflammatory and stress responses activate NF-κB in the hypothalamus and induce a signaling pathway that reduces production of gonadotropin-releasing hormone (GnRH) by neurons.
The case is particularly interesting when we realize that the aging phenotype can only be maintained by continuous activation of NF-κB. So here we have a multi-level interaction:
- Activation of NF-κB leads to
- Cellular aging, leading to
- Diminished production of GnRH, which then
- Acts (through cells with a receptor for it, or indirectly as a result of changes to GnRH-receptor-possessing cells) to decrease lifespan.
Cell energetics is not the solution, and will never lead to a solution because it makes the assumption that cells age. Cells take on the age-phenotype the body gives them.
Aging is not a defect – it’s a programmed progressive process, a continuation of development with the body doing more to kill itself with advancing years. Progressive life-states where each succeeding life-stage has a higher mortality (there are rare exceptions).
Cellular aging is externally controlled (cell non-autonomous). None of those remedies that slow ‘cell aging’ (basically all anti-aging medicines) can significantly extend anything but old age.
For change at the epigenomic/cellular level to travel up the biological hierarchy from cells to organ systems seems to take time. But the process can be repeated indefinitely (so far as we know).”
We may express concern about others. But each of us should also take responsibility for our own one precious life.













 Inauguration day
Inauguration day
 And 2021 will look like..?
And 2021 will look like..?
 Thanks for all the fish!
Thanks for all the fish!