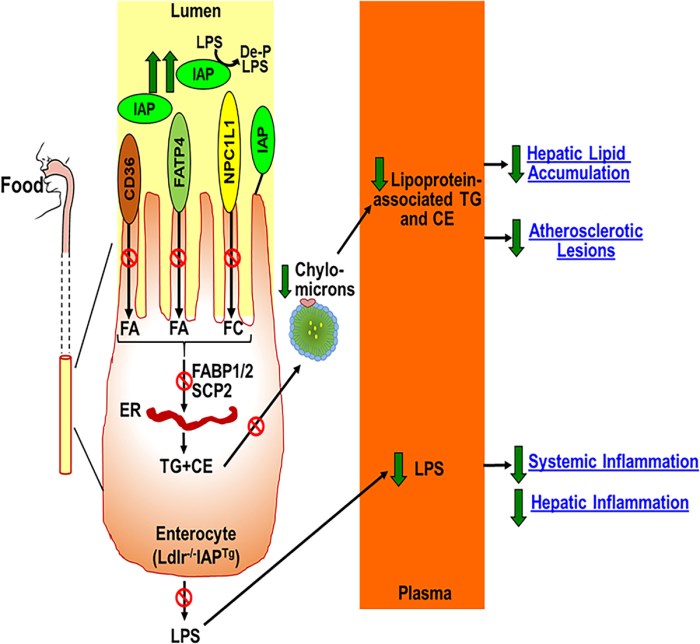
Three human studies investigated strains within microbiota species. The first from 2021 had obese child subjects:
“Dietary intervention is effective in human health promotion through modulation of gut microbiota. Diet can cause single-nucleotide polymorphisms (SNPs) to occur in gut microbiota, and some of these variations may lead to functional changes in human health.
Compared with normal diet, the WTP diet provided large quantities of whole-grain mix that was rich in starch, soluble and insoluble dietary fiber, protein, and amino acids, but contained a small amount of fat. When this excess and/or indigestible nutrition reached the colon, it brought environmental pressures to microbiota that stayed there.
This pressure could facilitate utilization of indigestible nutrition by causing microbial SNPs. Metabolic efficiencies of indigestible nutrition substrates would be enhanced to adapt to the shifted environment better.
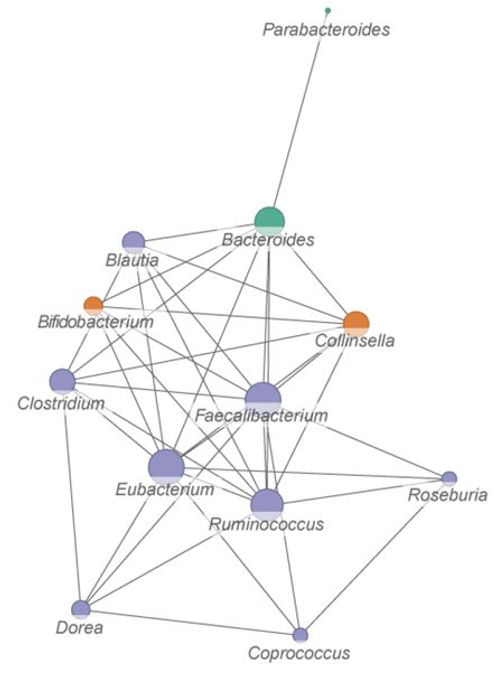
Although abundance of Bifidobacterium increased significantly by the intervention and became dominant strains responsible for nutrition metabolism, they had less BiasSNPs between the pre- and post-intervention group in comparison with Faecalibacterium. Finding F. prausnitzii as important functional strains influenced by intervention highlights the superiority of applying SNP analysis in studies of gut microbiota.
Though F. prausnitzii were well known for their biodiversity, we could not find functional reports about these SNPs. Future efforts are needed to verify/discern specific effects of these SNPs on encoded protein activity, their role on metabolism under high-fiber dietary intervention, and their potential beneficial or detrimental influences on host health.”
https://www.frontiersin.org/articles/10.3389/fmicb.2021.683714/full “Gut Microbial SNPs Induced by High-Fiber Diet Dominate Nutrition Metabolism and Environmental Adaption of Faecalibacterium prausnitzii in Obese Children”
A second 2021 human study investigated strain diversity in liver cirrhosis and Crohn’s disease:
“We constructed a computational framework to study strain heterogeneity in the gut microbiome of patients with liver cirrhosis (LC). Only Faecalibacterium prausnitzii showed different single-nucleotide polymorphism patterns between LC and healthy control (HC) groups.
Strain diversity analysis discovered that although most F. prausnitzii genomes are more deficient in LC group than in HC group at the strain level, a subgroup of 19 F. prausnitzii strains showed no sensitivity to LC, which is inconsistent with the species-level result.
More experiments need to be conducted so as to confirm the hypothesis of physiological differences among subgroups of F. prausnitzii strains. Our results suggest that strain heterogeneity should receive more attention.
With rapid development of sequencing technologies and experimental approaches, an increasing number of metagenomic studies will involve strain-level analysis. Such analysis of human metagenomes can help researchers develop more reliable disease diagnoses and treatment methods from a microbiological perspective.”
https://journals.asm.org/doi/10.1128/mSystems.00775-21 “Comprehensive Strain-Level Analysis of the Gut Microbe Faecalibacterium prausnitzii in Patients with Liver Cirrhosis”
A 2018 study investigated dietary fibers’ effects on Type 2 diabetics:
“In this study, we identified a group of acetate- and butyrate-producing bacterial strains that were selectively promoted by increased availability of diverse fermentable carbohydrates in the form of dietary fibers. These positive responders are likely key players for maintaining the mutualistic relationship between gut microbiota and the human host. Promoting this active group of SCFA producers not only enhanced a beneficial function but also maintained a gut environment that keeps detrimental bacteria at bay.
Only a small number of bacteria with genetic capacity for producing SCFAs were able to take advantage of this new resource and become dominant positive responders. The response, however, was strain specific: only one of the six strains of Faecalibacterium prausnitzii was promoted.

The 15 positive responders are from three different phyla, but they act as a guild to augment deficient SCFA production from the gut ecosystem by responding to increased fermentable carbohydrate availability in similar ways. When they are considered as a functional group, the abundance and evenness of this guild of SCFA producers correlate with host clinical outcomes.”
https://science.sciencemag.org/content/359/6380/1151.full “Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes”
These studies favored a prebiotic approach to make gut microbiota happy and reciprocal in human health. The second study investigated 135 known strains of F. prausnitzii, and the first study found beneficial F. prausnitzii strains not yet covered in genomic databases.
I found the first two studies by them citing the third. The third study was cited in Gut microbiota guilds.