Two 2023 reviews of Nrf2 and aging, starting with Nrf2-mitochondria interactions:
“We discuss molecular mechanisms of interactions between Nrf2 and mitochondria that influence the rate of aging and lifespan. Nrf2 activity positively affects both mitochondrial dynamics and mitochondrial quality control.
Nrf2 influences mitochondrial function through regulation of nuclear genome-encoded mitochondrial proteins and changes in the balance of ROS or other metabolites. In turn, multiple regulatory proteins functionally associated with mitochondria affect Nrf2 activity and even form mutual regulatory loops with Nrf2. These loops enable fine-tuning of cellular redox balance and, possibly, of the cellular metabolism as a whole.
mtDNA-encoded signal peptides interact with nuclear regulatory systems, first of all, Nrf2, and are possibly involved in regulation of the aging rate. Interactions between regulatory cascades that link programs ensuring maintenance of cellular homeostasis and cellular responses to oxidative stress are a significant part of both aging and anti-aging programs.
Understanding these interactions will be of great help in searching for molecular targets to counteract aging-associated diseases and aging itself. Future research on Nrf2 signaling and ability of various substances that activate the Nrf2 pathway to prevent age-associated chronic diseases will provide further insight into the role of Nrf2 activation as a possible longevity-promoting intervention.”
https://link.springer.com/article/10.1134/S0006297922120057 “Transcription Factor Nrf2 and Mitochondria – Friends or Foes in the Regulation of Aging Rate” (not freely available) Thanks to Dr. Gregory A. Shilovsky for providing a copy.
The second review evaluated whether Nrf2 is a master regulator of aging:
“This paper briefly presents main mechanisms of mammalian aging and roles of inflammation and oxidative stress in this process. Mechanisms of Nrf2 activity regulation, its involvement in aging and development of the senescence-associated secretory phenotype are also discussed.
The age-related decrease in Nrf2 activity is of universal interspecies character:
- Rodents with high Nrf2 activity have a longer lifespan than rodents with low activity.
- Genetic knockout of Nrf2 usually leads to the increased senescent phenotype in a variety of animal organs and tissues, and also reduces lifespan of female mice.
- There are also opposite examples, where Nrf2 knockout in aging mice reduced iron ions deposition in the brain, lowered the level of oxidative damage in the striatum, and also alleviated age-related motor dysfunction.
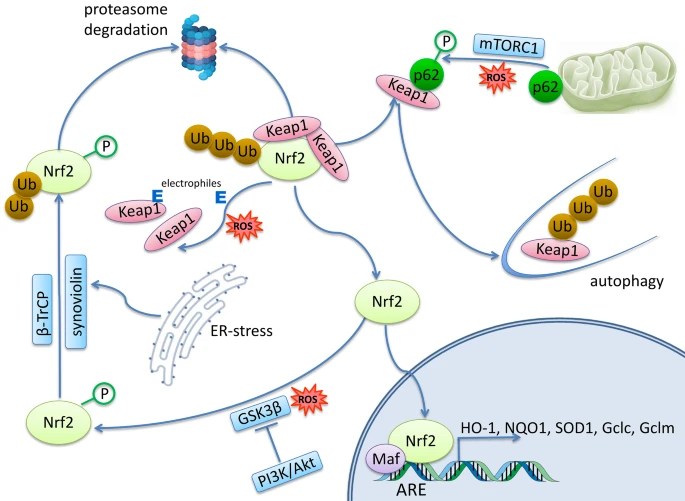
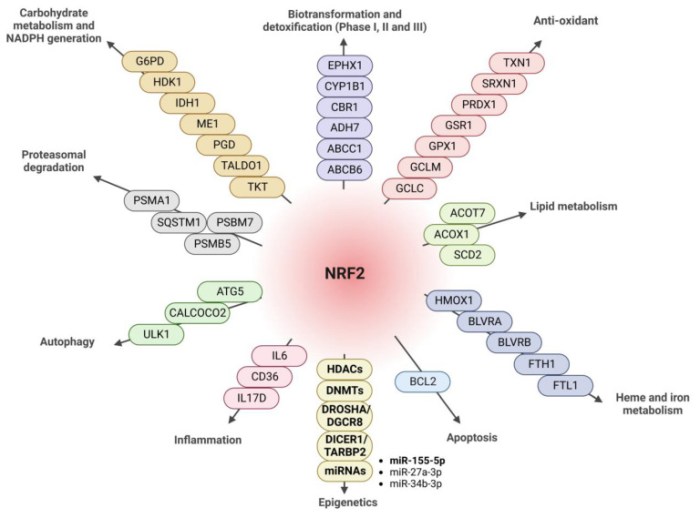
It would be incorrect to consider the effect of Nrf2 transcription factor at the organism level as exclusively antioxidant, anti-inflammatory, and, ultimately, anti-aging. Nrf2 controls many genes, products of which have complex, pleiotropic effects on the body:
- No experiments that use Nrf2 chemical inducers as anti-aging drugs have been performed so far.
- Nrf2 is not involved in life extension caused by caloric restriction.
- Epigenetic clocks do not reveal transcription factors activity of which changes with aging.
Aging is accompanied by changes in gene expression profiles, which are tissue- and species-specific. These changes only to a small extent include genes controlled by Nrf2. At the moment, it cannot be concluded that Nrf2 is the master regulator of the aging process.”
https://link.springer.com/article/10.1134/S0006297922120045 “Does Nrf2 Play a Role of a Master Regulator of Mammalian Aging?”