This week I dialed back eating microwaved 3-day-old broccoli / red cabbage / mustard sprouts from twice a day to once a day. For my reasoning, here are two papers on broccoli sprouts and thyroid function, with the first a 2018 human study:
“We analyzed biochemical measures of thyroid function and thyroid autoimmunity in a subset of participants in a broccoli sprout clinical trial. The present work is a retrospective analysis of a subset of serum samples collected during a clinical trial conducted from mid-October 2011 to early January 2012.
130 individuals received placebo beverage, and 137 received broccoli sprout beverage for 84 consecutive days (12 weeks). Blood samples from day 0 and day 84 were analyzed in a subset of 45 female participants (19 placebo, 26 broccoli sprout beverage) for serum thyroid-stimulating hormone (TSH), free thyroxine (fT4), thyroglobulin (TG), anti-TG, and anti-thyroid peroxidase (anti-TPO) antibodies.
The percentage of patients with subclinical hypothyroidism (elevated TSH with normal fT4) was not significantly different between the two groups either before or after treatment.
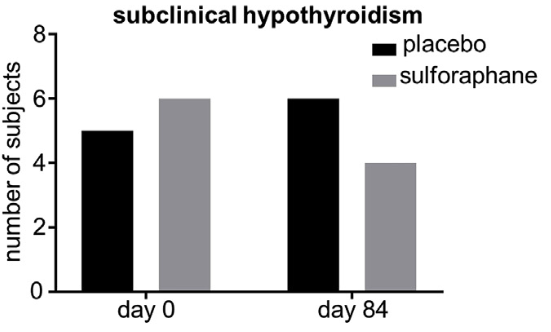
Daily ingestion of a broccoli sprout extract beverage over 84 days had no deleterious effect on thyroid function tests or measures of thyroid autoimmunity. It may be prudent to evaluate thyroidal safety of plant-based food supplements on a case-by-case basis.”
https://www.sciencedirect.com/science/article/abs/pii/S0278691519300547 “Broccoli sprout beverage is safe for thyroid hormonal and autoimmune status: Results of a 12-week randomized trial” (not freely available)
A 2020 review by three of these coauthors summarized further details:
“One difference between the thyroid and other tissues is that ROS are not primarily a byproduct of its physiology, but an indispensable part of it. Thyroid follicular cells actively produce H2O2 to facilitate a cascade of redox reactions that sequentially oxidize iodide, iodinate tyrosine residues within Tg, and couple iodinated tyrosine residues of Tg to each other to form T4 and T3 (triiodothyronine).
There exists a fail-safe mechanism in which specific combinations of four Keap1 cysteines can form a disulfide bond to sense H2O2. This sensing mechanism appears to be distinct from that triggered by other Nrf2 inducers, such as electrophiles.
Findings from Keap1KD mice suggest that chronic genetic activation of Nrf2 signaling may have negative consequences for the thyroid gland. However, analysis of data from a clinical trial has shown that consumption of a broccoli sprout beverage (yielding pharmacologically active amounts of the Nrf2-activating compound sulforaphane) is safe for thyroid hormonal and autoimmune status during a 12-week administration period.
Nevertheless, it appears prudent to monitor thyroid function and thyroid volume (at least by palpation) in patients treated with Nrf2-modulating compounds in clinical trials or clinical practice.”
https://www.mdpi.com/2076-3921/9/11/1082/htm “The Keap1/Nrf2 Signaling Pathway in the Thyroid—2020 Update”
My Day 70 lab results for inflammation markers were great:

A year later, IL-6 was below the test’s detection limit, and high-sensitivity C-reactive protein could hardly have been better at 0.24 mg/L.
But TSH (reference interval 0.45 – 4.50 μIU/mL) increased from 3.01 to 7.50. Here’s what Labcorp Technical Review L8186 said:
“The panel concluded that despite the fact that serum TSH concentrations higher than 2.5 μIU/mL but less than 4.5 μIU/mL may identify some individuals with the earliest stage of hypothyroidism, there is no evidence for associated adverse consequences. Additionally, consequences of subclinical hypothyroidism with serum TSH levels between 4.5 μIU/mL and 10 μIU/mL are minimal, and the panel recommends against routine treatment of patients with TSH levels in these ranges.”
I went in last weekend to retest. Although the provider verbally agreed to test TSH, free T3, and free T4, a different test was ordered.
TSH was still high at 5.85 μIU/mL. Other measurements (Total T4, T3 Uptake, and Free Thyroxine Index) aren’t suitable substitutes for free T3 and free T4. I’ll specify Labcorp test numbers next time.
My hypothesis is that preconditioning my endogenous ARE system twice daily worked alright elsewhere, but not for my thyroid. We’ll find out in 2022 whether halving the electrophilic activations of my Nrf2 signaling pathway has any effect on thyroid measurements.
I don’t take anything with, or an hour before or after these very reactive isothiocyanates. I continue to eat 3-day-old oat sprouts twice a day with other foods.