A trio of papers on Vitamin K2, the first being a 2021 review that emphasized dual effects:
“Osteoporosis (OP) is the most common bone disease that affects elderly men and women. It is a metabolic skeletal disorder caused by an imbalance between bone formation and resorption, leading to a loss of bone mass and quality, skeletal structure deterioration, and an increased risk of fractures.
Vascular calcification is defined as ectopic deposition of mineral matrix in vessel wall. It occurs prevalently in aging and primary chronic conditions (hypertension, diabetes mellitus, and chronic kidney disease), representing an important risk factor for cardiovascular morbidity and mortality.
Studies have provided support for a close link between bone and vascular health. Findings suggest that bone loss in OP may promote and increase the risk of cardiovascular events and vascular atherosclerosis.
Vitamin K2 is involved in a phenomenon in which a low calcium deposition in bone tends to be associated with a parallel increase of calcium deposition in vessel wall as a consequence of impaired calcium metabolism. Most production of Vitamin K2 in humans takes place in intestines. However, the amount derived from intestinal bacteria is poorly absorbed, and is not able to reach concentrations required to exert physiological functions.
Vitamin K2‘s ability to reduce loss of bone mineral density and fracture risk, as well as to improve bone quality, has been described by several clinical studies, which have confirmed that osteocalcin (OC) γ-carboxylation is the main mechanism of action through which this natural compound is able to improve bone health. Clinical evidence suggests an analogous protective role of Vitamin K2 at the vascular level, emphasizing a strict association between:
- Vitamin serum level;
- Matrix gla protein (MGP) γ-carboxylation levels;
- Reduction of vascular smooth muscle cells osteogenic trans-differentiation; and
- Possible risk of cardiovascular events.”
https://www.mdpi.com/2072-6643/13/4/1222/htm “The Dual Role of Vitamin K2 in ‘Bone-Vascular Crosstalk’: Opposite Effects on Bone Loss and Vascular Calcification”
A second 2021 review emphasized aging:
“Vitamin K can:
- Carboxylate OC (a protein capable of transporting and fixing calcium in bone);
- Activate MGP (an inhibitor of vascular calcification and cardiovascular events); and
- Carboxylate Gas6 protein (involved in brain physiology and a cognitive decline and neurodegenerative disease inhibitor).
By improving insulin sensitivity, Vitamin K lowers diabetes risk. It also exerts antiproliferative, proapoptotic, autophagic effects, and has been associated with a reduced risk of cancer.
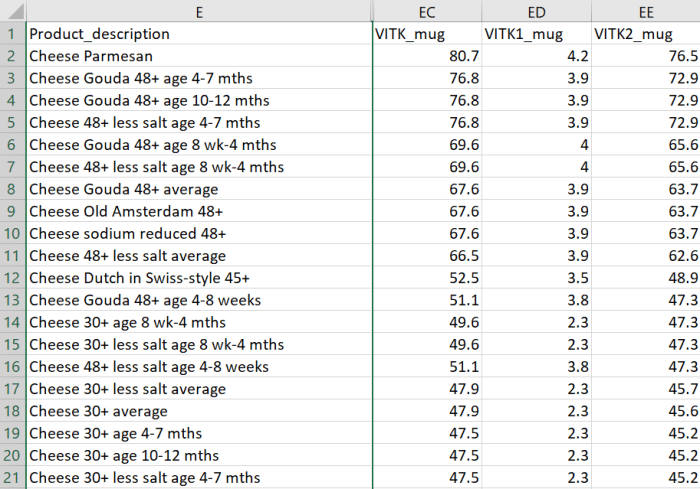
The most common [Vitamin K2] subtypes in humans are the short-chain MK[menaquinone]-4, which is the only MK produced by systemic conversion of phylloquinone [Vitamin K1] to menaquinone, and MK-7 through MK-10, which are synthesized by bacteria. The main sources of Vitamin K2 are fermented foods, cheeses, eggs, and meats.”
https://www.mdpi.com/2076-3921/10/4/566/htm “The Role of Vitamin K in Humans: Implication in Aging and Age-Associated Diseases”
The third paper – somehow not cited by these two reviews – was a 2006 human study that performed four experiments:
“The synthetic short-chain vitamin K1 is commonly used in food supplements, but recently the natural long-chain MK-7 has also become available as an over-the-counter supplement. The purpose of this paper was to compare in healthy volunteers absorption and efficacy of K1 and MK-7.
Serum vitamin K species were used as a marker for absorption and OC carboxylation as a marker for activity. Both K1 and MK-7 were absorbed well, with peak serum concentrations at 4 hours after intake.
A major difference was:
- Very long half-life time of MK-7, resulting in much more stable serum levels; and
- Accumulation of MK-7 to higher levels (7- to 8-fold) during prolonged intake.
MK-7 induced more complete carboxylation of OC.

Accumulation and efficacy of K vitamins during long-term daily administration. Participants received in a crossover design either K1 (○) or MK-7 (•) or placebo; in the latter case only K1 (▴) could be detected.
- (A) Circulating levels of vitamin K; baseline levels for K1 were subtracted; no MK-7 could be detected at baseline.
- (B) Ratio between circulating carboxylated and undercarboxylated osteocalcin (cOC/ucOC); at baseline the ratio was 1.74 for MK-7, 1.8 for K1, and 1.7 for the placebo group.
MK-7 accumulated during the first 2 weeks until it reached a plateau level of about 10 nM (6 μg/L), whereas K1 remained slightly above placebo values during the entire study period. Efficacy of both K vitamins for OC carboxylation was monitored using the ratio between circulating cOC and ucOC, and it turned out that within 3 days both vitamins had induced increased cOC.
But only by taking MK-7 did the effect continue to increase during the entire study period.
Taken together, these data demonstrate considerable differences between MK-7 and K1:
- Higher and more stable serum levels are reached with MK-7; and
- MK-7 has a higher efficacy in both hepatic and extrahepatic protein carboxylation.”
https://ashpublications.org/blood/article/109/8/3279/23729/Vitamin-K-containing-dietary-supplements “Vitamin K–containing dietary supplements: comparison of synthetic vitamin K1 and natto-derived menaquinone-7″
I’ve tried various things over the years to address hypertension. I stopped high blood pressure medications briefly to see if each intervention worked. They all haven’t, presumably because I didn’t address causes.
More recently, I broke my left big toe on furniture while walking around in the dark last month, and haven’t recovered. No pictures from walking on the beach at sunrise because it still isn’t possible. 😦
A link between these two health conditions could be Vitamin K2. I don’t eat fermented foods because of their high sodium, or dairy products, and haven’t supplemented Vitamin K2.
Next week I’ll start a 300 μg MK-7 daily dose. Current Vitamin D3 dose is 3800 IU, compared to the second paper of Part 2 of Vitamin K2 – What can it do? which is 400 μg MK-7 and 3200 Vitamin D3.