This 2018 French/Italian/Swiss rodent study used a prenatally restraint stressed (PRS) model to create problems that could be resolved by various chemicals:
“S 47445 is a positive modulator of glutamate AMPA-type receptors, possessing neurotrophic and enhancing synaptic plasticity effects as well as pro-cognitive and anti-stress properties.
Most of studies examining the antidepressant effects of new molecules are carried out using behavioral tests performed in unstressed animals.
Corticosterone-treated mice and rats exposed to chronic stress are models that do not recapitulate the early programming of stress-related disorders, which likely originates in the perinatal period. The PRS rat model is characterized by a prolonged corticosterone response to stress and by abnormal behavior.
All the behavioral alterations induced by PRS were corrected by chronic S 47445 administration at both doses.”
The paper included a section comparing S 47445 to ketamine:
“Ketamine, however, causes severe cognitive impairment and psychotomimetic [mimics the symptoms of psychosis, reference not freely available] effects that are direct consequences of the prolonged inhibition of NMDA receptors in cortical and hippocampal interneurons, and seriously limit the chronic administration of the drug in the clinical setting. [reference not freely available]
S 47445 by inducing a direct activation of AMPARs displayed an antidepressant activity without the adverse effect of ketamine. Indeed, contrary to ketamine, S 47445 presented no psychotomimetic effects and induced no occurrence of spontaneous epileptic seizures. [reference freely available] Moreover, S 47445 also presented pro-cognitive properties.”
Compare the above with this April 2018 Chicago Tribune story that had opinions with no linked references:
“ketamine, an anesthetic used to sedate both people and animals before surgery. It’s also a notorious street drug, abused by clubgoers seeking a trancelike, hallucinatory high. But in recent years, numerous studies have found that ketamine can be an effective and speedy treatment for people with depression.”
Which coverage better informed us?
Treating prenatal stress-related disorders with an oxytocin receptor agonist was performed by several of this paper’s coauthors. One references to it was:
“We have already reported that depolarization-evoked glutamate release in the ventral hippocampus is negatively correlated with risk-taking behavior of PRS rats, and that such correlation can be corrected by chronic treatment with monoaminergic/ melatoninergic antidepressants or oxytocin receptor agonist. Thus, an impairment of glutamatergic transmission in the ventral hippocampus lies at the core of the pathological phenotype of PRS rats.”
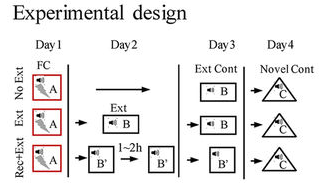
Looking at the above graphic of the experimental design, I’m not sure why the term perinatal (occurring during or pertaining to the phase surrounding the time of birth) was used in the paper’s title and content to describe the stress period. The pregnant females were stressed three times every day during the second half of pregnancy up until delivery, so the prenatal (previous to birth) term was more applicable.
So, how does this study help humans?
One takeaway is to avoid stressing pregnant mothers-to-be if her children will be expected to become adults without cognitive, emotional, and behavioral problems.
The study demonstrated one way prenatal events cause lifelong effects. The PRS model provides another example of why it’s useless to ask adult humans to self-report causes of epigenetic problems in their lives when these originated before birth, during infancy, or in early childhood, well before humans develop sufficient cognitive capability to recognize such situations. It’s incomprehensible that this unreliable paradigm is still given significant weight in stress studies, especially when experimental designs:
“Do not recapitulate the early programming of stress-related disorders, which likely originates in the perinatal period.”
Also, a relevant difference between humans and PRS rats is that we can ourselves individually change our responses to experiential causes of ongoing adverse effects. Standard methodologies can only apply external treatments such as those mentioned above.
https://www.sciencedirect.com/science/article/pii/S0028390818301291 “The reduction in glutamate release is predictive of cognitive and emotional alterations that are corrected by the positive modulator of AMPA receptors S 47445 in perinatal stressed rats” (not freely available) Thanks to coauthors Stefania Maccari and Dr. Jerome Mairesse for providing a copy.