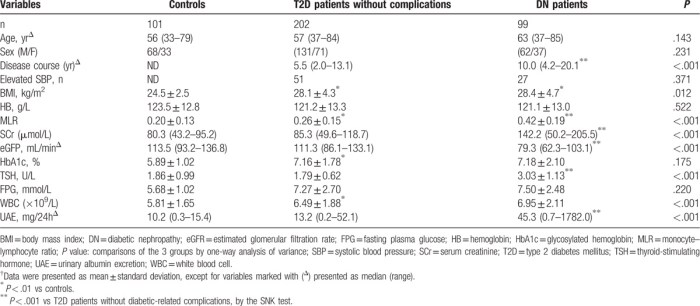
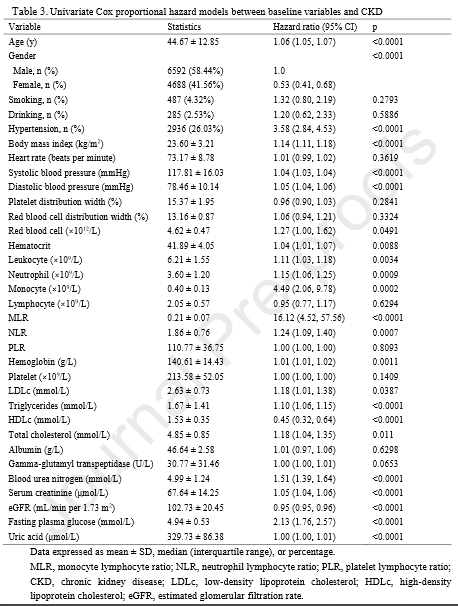
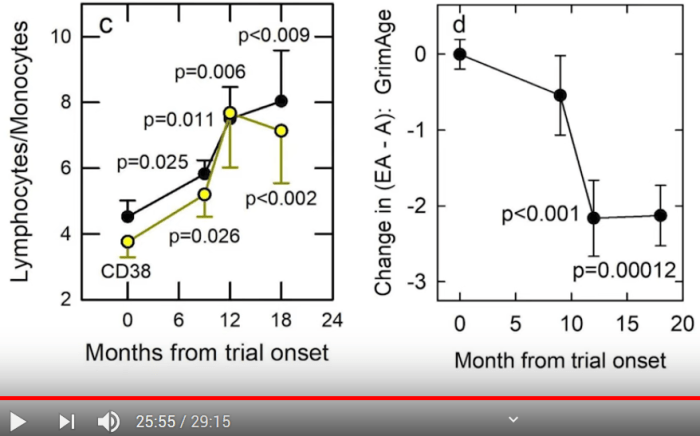
Two papers on broccoli compounds and gut microbiota relationships, with the first a 2021 article:
“We provide a supportive environment and a supply of nutrition and, in return, the microbiome delivers benefits to our health. What exactly are those benefits, and how can we maximise them?
Fibre component of food was thought to be completely indigestible roughage, but we now know that there is a digestible (a.k.a. soluble) component that can be fermented by bacteria resident in the large intestine, providing them with nutrition. There is also non-digestible fibre (a.k.a. insoluble fibre), which is not fermented by gut bacteria and includes plant cell walls formed from cellulose and lignin.
However, when cell walls remain intact, they encapsulate starch contained within cells and physically protect it from full digestion in the small intestine, ensuring that more passes into the large intestine where it can then be fermented by bacteria.
A bioactive is any chemical found in plant-based food that affects biological processes in the body, promoting better health or reducing risk of disease. Unlike macronutrients, such as carbohydrates and proteins, bioactive compounds are usually found in small amounts.
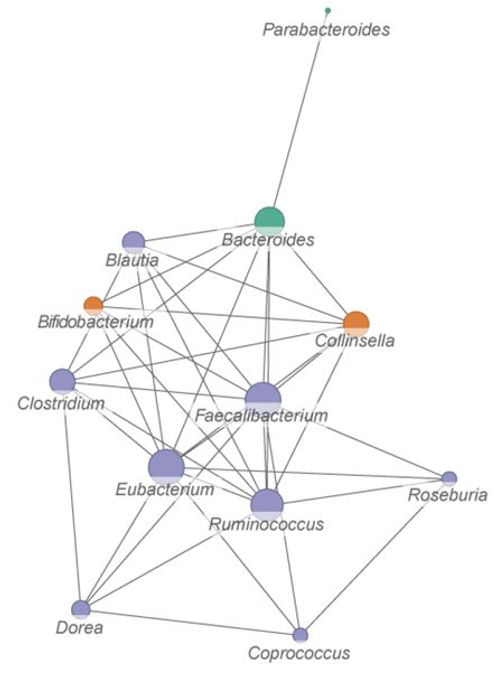
One class of bioactives where this has been known for some time is glucosinolates. For some compounds, including glucosinolates, we have identified particular bacteria that perform this task. For others, we still do not know which microbes are responsible.
S-methylcysteine sulphoxide (SMCSO) is found in brassicas but also in garlic and its relatives. Its metabolic breakdown products have been associated with protective effects against prostate and colon cancer, diabetes, and cardiovascular disease.
SMCSO-derived compounds are highly bioactive, so understanding how they affect the body’s central metabolic pathways could explain some of their health benefits. Only recently have we found clues to bacteria responsible.”
https://ifst.onlinelibrary.wiley.com/doi/10.1002/fsat.3501_6.x “Gut reaction”
The 2020 study cited for SMCSO was an in vitro 2020 study by their coworkers:
“We examined effects of a broccoli leachate (BL) on composition and function of human faecal microbiomes of five different participants under in vitro conditions. Bacterial isolates from these communities were then tested for their ability to metabolise glucosinolates and SMCSO.
We believe that this is the first study that shows reduction of dietary compound SMCSO by bacteria isolated from human faeces. Microbial communities cultured in vitro in BL media were observed to have enhanced growth of lactic acid bacteria, such as lactobacilli, with a corresponding increase in levels of lactate and short-chain fatty acids (SCFAs).
These results would have been strengthened by analysing soluble fibre content of BL media. As such, it is difficult to relate these results to in vivo SCFA production following consumption of broccoli.”
https://link.springer.com/article/10.1007/s00394-020-02405-y “Effects of in vitro metabolism of a broccoli leachate, glucosinolates and S-methylcysteine sulphoxide on the human faecal microbiome”
Which one of this pair is a male? I’ll guess on the right, as it subsequently turned to face me – a threat – when I walked passed them at a distance.