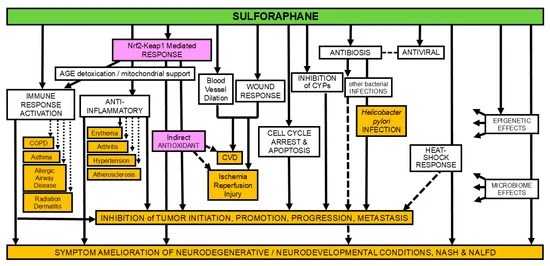
A 2025 review subject was taurine’s beneficial effects on mitochondria:
“Taurine has multiple and complex functions in protecting mitochondria against oxidative-nitrosative stress. We introduce a novel potential role for taurine in protecting from deuterium (heavy hydrogen) toxicity. This can be of crucial impact to either normal or cancer cells that have highly different mitochondrial redox status.
Deuterium is an isotope of hydrogen with a neutron as well as a proton, making it about twice as heavy as hydrogen. We first explain the important role that the gut microbiome and gut sulfomucin barrier play in deuterium management. We describe synergistic effects of taurine in the gut to protect against deleterious accumulation of deuterium in mitochondria, which disrupts ATP synthesis by ATPase pumps.
Taurine’s derivatives, N-chlorotaurine (NCT) and N-bromotaurine (NBrT), produced through spontaneous reaction of taurine with hypochlorite and hypobromite, have fascinating regulatory roles to protect from oxidative stress and beyond. We describe how taurine could potentially alleviate deuterium stress, primarily through metabolic collaboration among various gut microflora to produce deuterium depleted nutrients and deuterium depleted water (DDW), and in this way protect against leaky gut barrier, inflammatory bowel disease, and colon cancer.
Taurine cannot be metabolized by human cells, but gut microbes are able to break it down to release sulfite, which then gets oxidized to sulfate anions that become available to support synthesis of sulfomucins. Taurine protects against many diseases linked to mitochondrial defects, such as aging, metabolic syndrome, cancer, cardiovascular diseases and neurological disorders.
We present a novel view that gut microbes play an essential role in providing deuterium depleted (deupleted) nutrients, especially, butyrate, to the host colonocytes forming the gut barrier. We propose that sulfomucins synthesized by goblet cells not only protect the barrier from pathogens, but also trap and sequester deuterium, thus reducing mitochondrial deuterium levels, resulting in improved mitochondrial health.
Due to taurine, redox buffer glutathione (GSH) further stabilizes the membrane potential. GSH not only reduces radical oxygen species (ROS) during oxidative stress, but it also assists in production of deupleted water in mitochondria.
Spontaneous oxidation of two GSH molecules to produce GSSG in the presence of hydrogen peroxide yields two molecules of DDW. Just as for glutathione, bilirubin can produce DDW indefinitely through chronic recycling between bilirubin and biliverdin, capturing a deupleted proton in NADPH to produce a DDW molecule in each cycle.
A novelty that arises from this investigation is introduction of the role that deuterium plays in mitochondrial disease, and ways in which taurine may facilitate maintenance of low deuterium in mitochondrial ATPase pumps. Excess deuterium causes a stutter in the pumps, which leads to inefficiencies in ATP production and an increase in ROS.”
https://pmc.ncbi.nlm.nih.gov/articles/PMC11717795/ “Taurine prevents mitochondrial dysfunction and protects mitochondria from reactive oxygen species and deuterium toxicity”