Here’s a 2025 review where the lead author is a retired researcher whose words readers might interpret as Science. As a reminder, unlike study researchers, reviewers are free to:
- Express their beliefs as facts;
- Over/under emphasize study limitations; and
- Disregard and misrepresent evidence as they see fit.
Reviewers also aren’t obligated to make post-publication corrections for their errors and distortions. For examples:
1. After the 7. Conclusions section, there’s an 8. Afterword: I3C and DIM section. The phrase “As detailed in our earliest work on broccoli sprouts..” indicated a belief carried over from last century of the low importance of those research subjects.
Then, contrary to uncited clinical trials such as Our model clinical trial for Changing to a youthful phenotype with broccoli sprouts and Eat broccoli sprouts for DIM, “Broccoli sprouts had next to no indole glucosinolates.” And in the middle of downplaying I3C and DIM research, they stated: “There are 149 clinical studies on DIM and 11 on I3C listed on clinicaltrials.gov, suggesting a good safety profile. Potential efficacy and mode of action in humans are a subject of intense current investigation, though definitive answers will not come for some time.” 🧐
2. In the 3. Sulforaphane section, they asserted: “Glucosinolates such as glucoraphanin are ‘activated’ or converted to isothiocyanates such as sulforaphane by an enzyme called myrosinase, which is present in that same plant tissue (e.g., seed, sprout, broccoli head, or microgreen) and/or in bacteria that all humans possess in their gastrointestinal tracts.” and cited a 2016 book they coauthored that I can’t access.
The first 2021 paper of Broccoli sprout compounds and gut microbiota didn’t assert that “all humans” had certain gut microbiota that converted glucosinolates to isothiocyanates. That paper instead stated: “Human feeding trials have shown inter-individual variations in gut microbiome composition coincides with variations in ITC absorption and excretion, and some bacteria produce ITCs from glucosinolates.”
3. Nearly half of their cited references were in vitro cancer papers. I rarely curate those types of studies because of their undisclosed human-irrelevant factors. For example, from the second paper of Polyphenol Nrf2 activators:
Bioavailability studies reveal that maximum concentrations in plasma typically do not exceed 1 µM following consumption of 10–100 mg of a single phenolic compound, with the maximum concentration occurring typically less than 2 h after ingestion, then dropping quickly thereafter. In the case of the in vitro studies assessed herein, and with few exceptions, most of the studies employed concentrations >10 µM with some studies involving concentrations in the several hundred µM range, with the duration of exposure typically in the range of 24–72 h, far longer duration than the very short time interval of a few minutes to several hours in human in vivo situations.
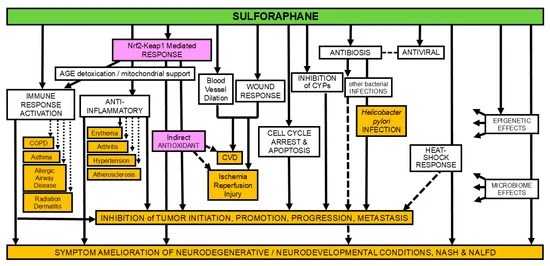
https://www.mdpi.com/2076-3417/15/2/522 “The Impact of Sulforaphane on Sex-Specific Conditions and Hormone Balance: A Comprehensive Review”