Two 2026 papers, with the first an in vitro study of over 2000 compounds to select those that best inhibit BACH1:
“BACH1 regulates the cellular oxidative stress responses by suppressing expression of cytoprotective genes. Dysregulated BACH1 activity has been implicated in a range of pathologies, including chronic inflammatory diseases, fibrosis, and cancer, making it a promising therapeutic target.
We identified four structurally distinct compounds that robustly inhibit BACH1 function. Notably, these compounds simultaneously activate transcription factor NRF2, suggesting the potential for a broader modulation of oxidative stress pathways.
However, while NRF2 induces expression of genes that protect against oxidative stress and inflammation and suppress ferroptosis, BACH1 represses them. While NRF2 broadly activates cytoprotective genes, BACH1 inhibition triggers a more restricted response but with a strong upregulation of HMOX1 (significantly stronger than the one obtained upon NRF2 activation).
As such, combined NRF2 activation and BACH1 inactivation is expected to produce a more potent antioxidant and anti-inflammatory effect than targeting either factor alone. Moreover, BACH1 regulates unique targets not shared with NRF2 and can dominantly repress genes even in the presence of active NRF2. This confers BACH1 inhibition distinct therapeutic value, particularly in contexts such as cancer cell invasion, where its suppression yields anti-metastatic effects.
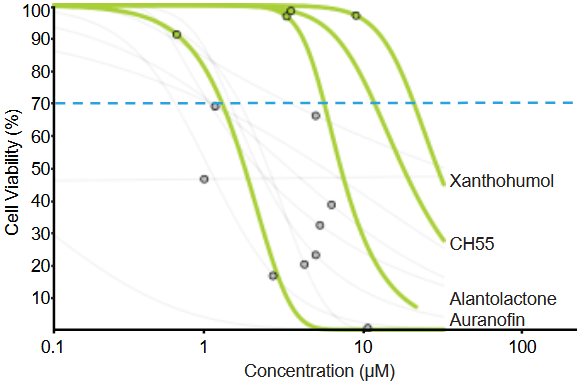
- Auranofin is an FDA-approved gold salt used in rheumatoid arthritis whose primary mechanism of action is inhibition of thioredoxin reductases (TrxRs).
- Xanthohumol is a natural compound, prenylated chalcone, that belongs to the flavonoid family, with reported antimicrobial, anti-inflammatory, and antioxidant activities, and demonstrated safety in phase I and phase II trials.
- Alantolactone is a natural compound, member of the sesquiterpene lactone class with anti-inflammatory and antioxidant effects, and has been tested in several animal models without reported toxicity.
- CH55 is a synthetic retinoid with high affinity for RAR-α and RAR-β and antifibrotic activity and has not been yet tested in vivo.
In summary, this work establishes a robust screening platform for identification of functional BACH1 inhibitors, and provides new chemical scaffolds with potential for future therapeutic development.”
https://papers.ssrn.com/sol3/papers.cfm?abstract_id=6439073 “Development of a High-Throughput Screening Platform for Identification of Functional BACH1 Inhibitors Reveals Compounds with Anti-Invasive Potential”
Xanthohumol is ninth on the Nrf2 activator list. BACH1 interactions were also covered in Part 3 of Broccoli sprouts activate the AMPK pathway.
A 2026 paper that was too recent to be referenced in the above study described two xanthohumol clinical trials. The first trial was designed to assess bioavailability in healthy people (6 men and 6 women), and the second was designed to determine bioactivity in 16 healthy women:
“The aim of the present project was to systematically investigate bioavailability of native xanthohumol compared to micellar xanthohumol at two doses (86 mg vs. 172 mg) in a randomized crossover trial. We furthermore examined short-term effects of xanthohumol on resting energy expenditure (REE), blood pressure (BP), and heart rate (HR) in a randomized placebo-controlled crossover study.
Micellar solubilization significantly increased area under the curve (AUC), maximum plasma concentration of xanthohumol (Cmax), timepoint of maximum plasma concentration of xanthohumol (tmax), and apparent bioavailability compared to native xanthohumol. The dose also significantly influenced plasma kinetics, but apparent bioavailability and tmax were dose-independent in contrast to AUC and Cmax. In our subsequent study, xanthohumol did not affect REE, substrate oxidation, BP, or HR.
Two properties of xanthohumol impair its bioavailability. First, xanthohumol is relatively unstable in an acidic environment, and second, xanthohumol is highly lipophilic and hydrophobic, insoluble in the aqueous environment of the intestinal lumen, and poorly absorbed into enterocytes.
In addition to determining typical plasma kinetic parameters (e.g., Cmax, tmax, and AUC), we also calculated the amount of xanthohumol absorbed using maximum plasma xanthohumol concentration. These estimated amounts are minimum quantities of xanthohumol that had to be absorbed to achieve observed plasma xanthohumol concentrations.
Results of these calculations emphasized the relatively poor bioavailability of xanthohumol in humans. Only ∼0.1% and ∼1.2% of native and micellar xanthohumol were absorbed, respectively.
However, a limitation of this plasma estimation is that fractional absorption of xanthohumol was underestimated because distribution of xanthohumol in cells and tissues was not taken into account. It is likely that more xanthohumol was actually absorbed. Whether the bioavailability of xanthohumol is sufficient for physiological efficacy must be investigated further in humans.
In conclusion, oral bioavailability of micellar xanthohumol was higher than that of native xanthohumol. Systemic availability of xanthohumol did not differ between men and women. Our study provides no evidence that xanthohumol acutely affects REE, BP, and HR.”
https://onlinelibrary.wiley.com/doi/10.1002/mnfr.70413 “The Bioavailability of Xanthohumol in Humans and the Influence of Formulation and Dose: Randomized Controlled Trial Data”